Unit 1: Sensory Systems
Learning Outcomes
Parts 1-4: Overall Organization of the Sensory Systems, Gustation, Olfaction
At the end of this part of this unit, you should be able to:
I. List the five main categories of sense receptors in the body based on the types of stimuli they respond to.
II. Describe the structure and function of cutaneous sensors and proprioceptors.
III. Describe the structure and function of the olfactory system.
IV. Describe the structure and function of the gustatory system.
Part 5: Audition and Balance
At the end of this part of this unit, you should be able to:
V. Describe the structure and functions of the external, middle and inner ear.
VI. Describe the physiology of hearing.
VII. Describe the physiology of static and dynamic balance.
Part 6: Vision
At the end of this part of this unit, you should be able to:
VIII. Identify the location and explain the function of each of the main components of the human eye.
IX. Describe the formation of an image on the retina.
X. Describe the overall distribution and functions of the two main types of photoreceptors in the retina.
XI. Describe the pathway of the nervous impulses from the photoreceptors of the retina to the brain.
Part 1: Overall Organization of the Sensory Systems
A major role of sensory receptors is to help us learn about the environment around us, or about the state of our internal environment. Stimuli from varying sources, and of different types, are received and changed into the electrochemical signals of the nervous system. This occurs when a stimulus changes the cell membrane potential of a sensory cell. The stimulus can cause the sensory cell to produce an action potential which is relayed into the central nervous system (CNS), where it is integrated with other sensory information—or sometimes higher cognitive functions—to become a conscious perception of that stimulus. The central integration in the CNS may then lead to a motor response.
Describing sensory function with the term sensation vs. perception is a deliberate distinction. Sensation is the activation of sensory receptor cells at the level of the stimulus. Perception is the central processing of sensory stimuli into a meaningful pattern. Perception is dependent on sensation, but not all sensations are perceived. Receptors are the cells or structures that detect sensations. A receptor cell is changed directly by a stimulus. A transmembrane protein receptor is a protein in the cell membrane which mediates a physiological change in a cell, most often through the opening of ion channels or changes in the cell signaling processes. Transmembrane receptors are activated by chemicals called ligands. For example, a molecule in food can serve as a ligand for taste receptors. Other transmembrane proteins, which are not accurately called receptors, are sensitive to mechanical or thermal changes. Physical changes in these proteins increase ion flow across the membrane, and can generate an action potential or a graded potential in the sensory neurons.
Sensory Receptors
Stimuli in the environment activate specialized receptor cells in the peripheral nervous system. Different types of stimuli are sensed by different types of receptor cells. Receptor cells can be classified into types on the basis of three different criteria: cell type, position, function. Receptors can be classified structurally on the basis of cell type and their position in relation to stimuli they sense. They can also be classified functionally on the basis of the transduction of stimuli, or how a mechanical stimulus, light, or chemical changes the cell membrane potential.
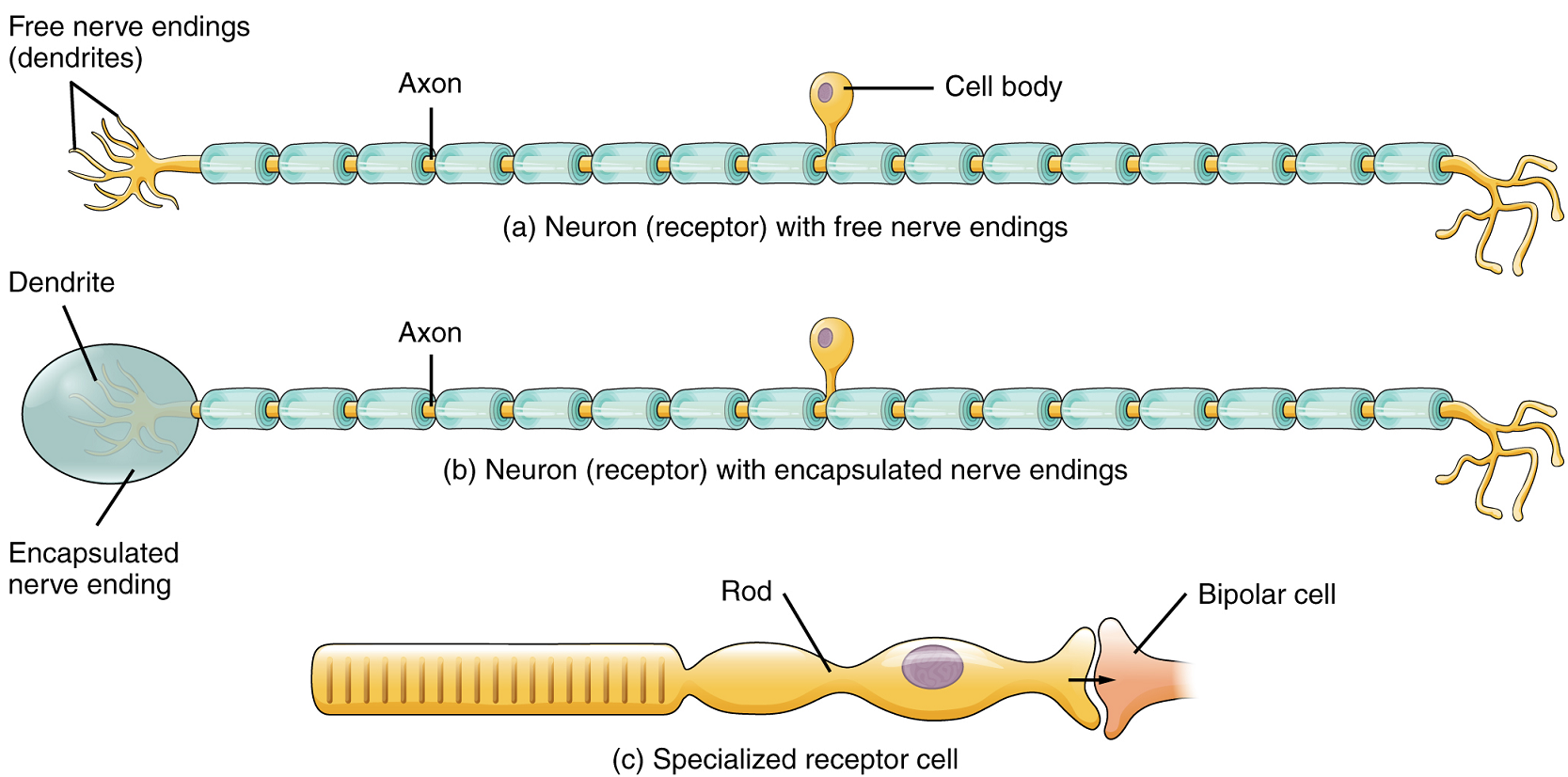
Structural Receptor Types: The cells that interpret information about the environment can either be: (1) a neuron that has a free nerve ending, with dendrites embedded in tissue that receives a sensation; (2) a neuron that has an encapsulated ending in which the sensory nerve endings are encapsulated in connective tissue that enhances their sensitivity; or (3) a specialized receptor cell, which has distinct structural components that interpret a specific type of stimulus (Figure 1). The pain and temperature receptors in the dermis of the skin are examples of neurons that have free nerve endings. Also located in the dermis of the skin are lamellated corpuscles, neurons with encapsulated nerve endings that respond to pressure and touch. The cells in the retina that respond to light stimuli are an example of a specialized receptor, in this case a photoreceptor.
Another way to classify receptors is based on the location of the stimuli to which they respond. An exteroceptor is a receptor that is located near a stimulus in the external environment, such as the somatosensory receptors that are located in the skin. An interoceptor is one that interprets stimuli from internal organs and tissues, such as the receptors that sense the increase in blood pressure in the aorta or carotid sinus. Finally, a proprioceptor is a receptor located near a moving part of the body, such as a muscle, which interprets the positions of the tissues as they move.
Functional Receptor Types: A third classification of receptors is based on how the receptor transduces stimuli into changes in membrane potential. Stimuli are of three general types. Some stimuli are ions and macromolecules that affect transmembrane receptor proteins when these chemicals diffuse across the cell membrane. Some stimuli are physical variations in the environment that affect receptor cell membrane potentials. Other stimuli include the electromagnetic radiation from visible light. For humans, the only electromagnetic energy that is perceived by our eyes is visible light. Other organisms have sensory receptors that humans lack, such as the heat sensors of snakes, the ultraviolet light sensors of bees, or the magnetic receptors in migratory birds.
Receptor cells can be further categorized on the basis of the type of stimuli they respond to. Chemical stimuli can be interpreted by a chemoreceptor which detects chemical stimuli that arise from the external environment, such as the compounds that determine an object’s taste or smell, or from the internal environment to monitor physiologically important parameters or the presence of damage. Osmoreceptors, for example, respond to solute concentrations of body fluids. The sensation of pain is primarily the result of the presence of chemicals released as a result of tissue damage or other intense stimuli through nociceptors.
Physical stimuli such as pressure and vibration, as well as the sensation of sound and body position (balance), are interpreted through mechanoreceptors. A physical stimulus which has its own type of receptor is temperature, sensed through thermoreceptors which are sensitive to temperatures either above (warmth/heat) or below (coolness/cold) normal body temperature.
Finally, highly specialized receptor cells called photoreceptors are used to detect light. They are found in the retina of the eye.
Sensory Modalities
Ask anyone what the senses are, and they are likely to list taste, smell, touch, hearing and sight. However, these are not all of the senses. The most obvious omission from this list is balance. Also, what is colloquially referred to as touch is a collection of senses that include pressure, vibration, stretch, and hair follicle position, on the basis of the type and location of mechanoreceptors that perceive these stimuli. Other often-overlooked senses include temperature perception by thermoreceptors and pain perception by nociceptors.
Within the realm of physiology, senses can be classified as either general or specific. A general sense is one that is distributed throughout the body and has receptor cells within the structures of other organs. Mechanoreceptors in the skin, muscles, or the walls of blood vessels are examples of receptor cells which function in this type of sense. General senses often contribute to the sense of touch, or to proprioception and kinesthesia (body movement), or to a visceral sense, which is most important to autonomic functions. A special sense is one that has a specific organ devoted to it, namely the eye, inner ear, tongue, or nose.
Each sense is referred to as a sensory modality. Modality refers to the way information is encoded, which is similar to the idea of transduction. The main sensory modalities can be described on the basis of how each is transduced. The chemical senses are taste and smell. The general sense which is usually referred to as touch includes chemical sensation in the form of nociception, or pain. Pressure, vibration, muscle stretching and the movement of hair by an external stimulus are all sensed by mechanoreceptors. Hearing and balance are also achieved through the actions of mechanoreceptors in sense organs. Finally, vision involves the activation of photoreceptors.
Listing all the different sensory modalities, which can number as many as 17, involves separating the major senses into more specific categories, or submodalities, of the larger sense. An individual sensory modality represents the sensation of a specific type of stimulus.
Part 2: Somatosensation
Somatosensation is the group of sensory modalities which is associated with touch, proprioception and interoception. These modalities include pressure, vibration, light touch, tickle, itch, temperature, pain and kinesthesia. Somatosensation is considered a general sense because somatosensory receptors are not associated with a specialized organ, but are instead spread throughout the body in a variety of organs. Many of these receptors are located in the skin and are referred to as cutaneous receptors. However, somatosensory receptors are also found in muscles, tendons, joint capsules, ligaments and in the walls of visceral organs.
Two types of somatosensory signals are transduced by free nerve endings: pain and temperature. These two modalities use thermoreceptors and nociceptors to transduce temperature and pain stimuli, respectively. Thermoreceptors are stimulated when local temperatures differ from body temperature. Some thermoreceptors are sensitive to just cold and others to just heat. Nociception is the sensation of potentially damaging stimuli, leading to pain. Mechanical, chemical or thermal stimuli beyond a set threshold will elicit painful sensations. Stressed or damaged tissues release chemicals which activate receptor proteins in the nociceptors. For example, the sensation of heat associated with spicy foods involves capsaicin, the active molecule in hot peppers. Capsaicin molecules bind to a particular type of transmembrane ion channel in nociceptors which is sensitive to temperatures above 37°C. The dynamics of capsaicin binding with this transmembrane ion channel is unusual in that the molecule remains bound for a long time. During this time, the ability of other stimuli to elicit pain sensations through the activated nociceptor will be diminished. For this reason, capsaicin can be used as a topical analgesic, such as in the product Icy Hot™.
If you drag your finger across a textured surface, the skin of your finger will vibrate. These low-frequency vibrations are sensed by mechanoreceptors called Merkel cells, also known as type I cutaneous mechanoreceptors. Merkel cells are located in the stratum basale of the epidermis. Deep pressure and vibration are transduced by lamellated (Pacinian) corpuscles, which are encapsulated receptors found deep in the dermis, or in the subcutaneous tissue. Light touch is transduced by the encapsulated nerve endings known as tactile (Meissner) corpuscles. Follicles are also wrapped in a plexus of nerve endings known as the hair follicle plexus. These nerve endings detect the movement of hair at the surface of the skin, such as when an insect is walking along the skin. Stretching of the skin is transduced by stretch receptors known as bulbous corpuscles. Bulbous corpuscles are also known as Ruffini corpuscles, or type II cutaneous mechanoreceptors.
Other somatosensory receptors are found in the joints and muscles, the proprioceptors. Stretch receptors monitor the stretching of tendons, muscles and the components of joints. Have you ever stretched your muscles before or after exercise and noticed that you can only stretch so far before your muscles spasm back to a less stretched state? This spasm is a reflex which is initiated by stretch receptors to avoid muscle tearing. These stretch receptors can also prevent the over-contraction of a muscle. In skeletal muscle tissue, these stretch receptors are called muscle spindles. Golgi tendon organs similarly transduce the stretch levels of tendons. Bulbous corpuscles are also present in joint capsules, where they measure stretch in the components of the skeletal system within the joint.
The main types of nerve endings, their locations and the stimuli they transduce are presented in Table 1.
| Name | Historical (eponymous) name | Location(s) | Stimuli |
|---|---|---|---|
| Free nerve endings | – | Dermis, cornea, tongue, joint capsules, visceral organs | Pain, temperature, mechanical deformation |
| Mechanoreceptors | Merkel’s discs | Epidermal-dermal junction, mucosal membranes | Low frequency vibration (5-15 Hz) |
| Bulbous corpuscle | Ruffini’s corpuscle | Dermis, joint capsultes | Stretch |
| Tactile corpuscle | Meissner’s corpuscle | Papillary dermis, especially in the fingertips and lips | Light touch, vibrations below 50 Hz |
| Lamellated corpuscle | Pacinian corpuscle | Depp dermis, subcutaneous tissue | Deep pressure, high-frequency vibration (~250 Hz) |
| Hair follicle plexus | – | Wrapped around hair follicles in the dermis | Movement of hair |
| Muscle spindle | – | In line with skeletal muscle fibers | Muscle contraction & stretch |
| Tendon stretch organ | Golgi tendon organ | In line with tendons | Stretch of tendons |
Part 3: Gustation
Only a few recognized submodalities exist within the sense of taste, or gustation. Until the mid-1980s, only four tastes were recognized: sweet, salty, sour and bitter. Further research led to recognition of the fifth taste: umami. Umami is a Japanese word which means “delicious taste,” and is often translated to mean savory. Very recent research has suggested that there may also be a sixth taste for fats, or lipids.
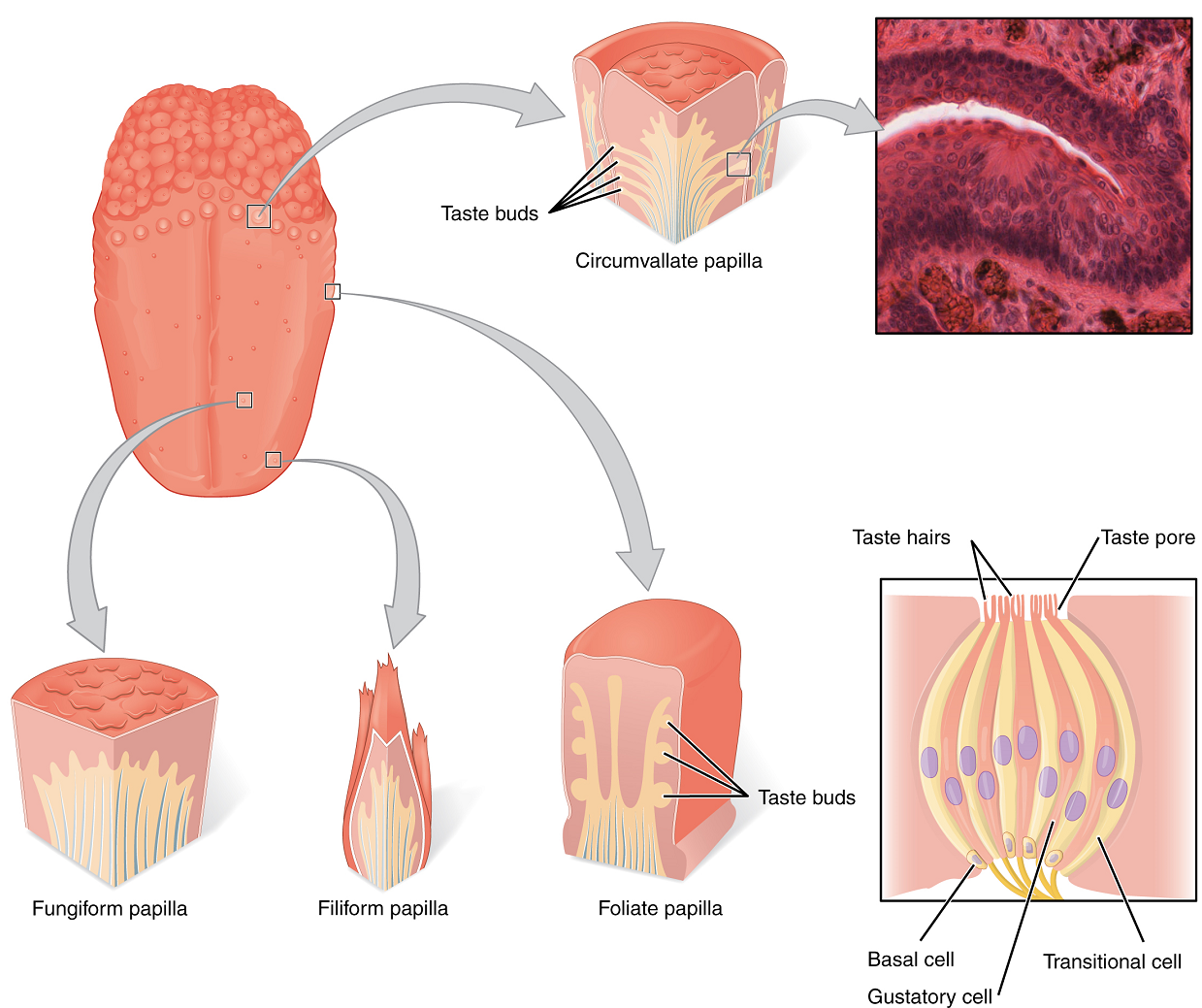
Gustation is the special sense associated with the tongue. The surface of the tongue, along with the rest of the oral cavity, is lined by a stratified squamous epithelium. Raised bumps called papillae (singular = papilla) contain the structures for gustatory transduction (Figure 2). Within the papillae are taste buds which contain specialized gustatory receptor cells for the transduction of taste stimuli. These receptor cells are sensitive to the chemicals contained within ingested foods.
Salty taste is simply the perception of sodium ions (Na+) in the saliva. When you eat something salty, the salt crystals dissociate into the component ions Na+ and Cl–, which dissolve into the saliva in your mouth. The Na+ concentration becomes high outside the gustatory cells, creating a strong concentration gradient that drives the diffusion of the ion into the cells. The entry of Na+ into these cells results in the depolarization of the cell membrane and the generation of a receptor potential.
Sour taste is the perception of H+ concentration. There are insufficient hydrogen ions (protons) in the extracellular fluid to depolarize the taste cell on their own – remember that even low concentrations of protons taste very sour; hence, the protons depolarize the cell by increasing sodium ion influx and decreasing potassium ion efflux. Sour flavors are, essentially, the perception of acids in our food. Increasing hydrogen ion concentrations in the saliva (lowering saliva pH) triggers progressively stronger graded potentials in the gustatory cells. For example, orange juice—which contains citric acid—will taste sour because it has a pH value of approximately 3. Of course, it is often sweetened so that the sour taste is masked.
The first two tastes (salty and sour) are triggered by the cations Na+ and H+. The other tastes result from food molecules binding to a G protein–coupled receptor. A G protein signal transduction system ultimately leads to depolarization of the gustatory cell. The sweet taste is the sensitivity of gustatory cells to the presence of glucose dissolved in the saliva. Other monosaccharides such as fructose, or artificial sweeteners such as aspartame (NutraSweet™), saccharine, or sucralose (Splenda™) also activate the sweet receptors. The affinity for each of these molecules varies, and some will taste sweeter than glucose because they bind to the G protein–coupled receptor differently.
Bitter taste is similar to sweet in that food molecules bind to G protein–coupled receptors. However, there are a number of different ways in which this can happen because there are a large diversity of bitter-tasting molecules. Some bitter molecules depolarize gustatory cells, whereas others hyperpolarize gustatory cells. Likewise, some bitter molecules increase G protein activation within the gustatory cells, whereas other bitter molecules decrease G protein activation. The specific response depends on which molecule is binding to the receptor.
One major group of bitter-tasting molecules are alkaloids. Alkaloids are nitrogen containing molecules that are commonly found in bitter-tasting plant products, such as coffee, hops (in beer), tannins (in wine), tea, and aspirin. By containing toxic alkaloids, the plant is less susceptible to microbe infection and less attractive to herbivores.
Therefore, the function of bitter taste may primarily be related to stimulating the gag reflex to avoid ingesting poisons. Because of this, many bitter foods that are normally ingested are often combined with a sweet component to make them more palatable (cream and sugar in coffee, for example). The highest concentration of bitter receptors appear to be in the posterior tongue, where a gag reflex could still spit out poisonous food.
The taste known as umami is often referred to as the savory taste. Like sweet and bitter, it is based on the activation of G protein–coupled receptors by a specific molecule. The molecule that activates this receptor is the amino acid L-glutamate. Therefore, the umami flavor is often perceived while eating protein-rich foods. Not surprisingly, dishes that contain meat are often described as savory.
Once the gustatory cells are activated by taste molecules through a change in the membrane potential (usually a depolarization causing a graded potential), these cells release neurotransmitters onto the dendrites of sensory neurons. These neurons are part of the facial and glossopharyngeal cranial nerves, as well as a component of the vagus nerve dedicated to the gag reflex. The facial nerve connects with taste buds in the anterior third of the tongue. The glossopharyngeal nerve connects with taste buds in the posterior two thirds of the tongue and the pharynx. The vagus nerve connects with taste buds in the extreme posterior of the tongue and the pharynx. Sensory neurons in these cranial nerves carry gustatory information to the solitary nucleus of the medulla oblongata, where they synapse with other neurons carrying the information to the thalamus. Axons from thalamic neurons then project to the primary gustatory cortex of the cerebrum, where taste is processed and consciously perceived. Other pathways also bring the information to the limbic system and hypothalamus, which are involved in emotional responses elicited by tasting food. Finally, the information passing through the medulla oblongata also triggers reflexes which contribute to digestion by increasing the secretion of saliva and gastric juices.
Part 4: Olfaction
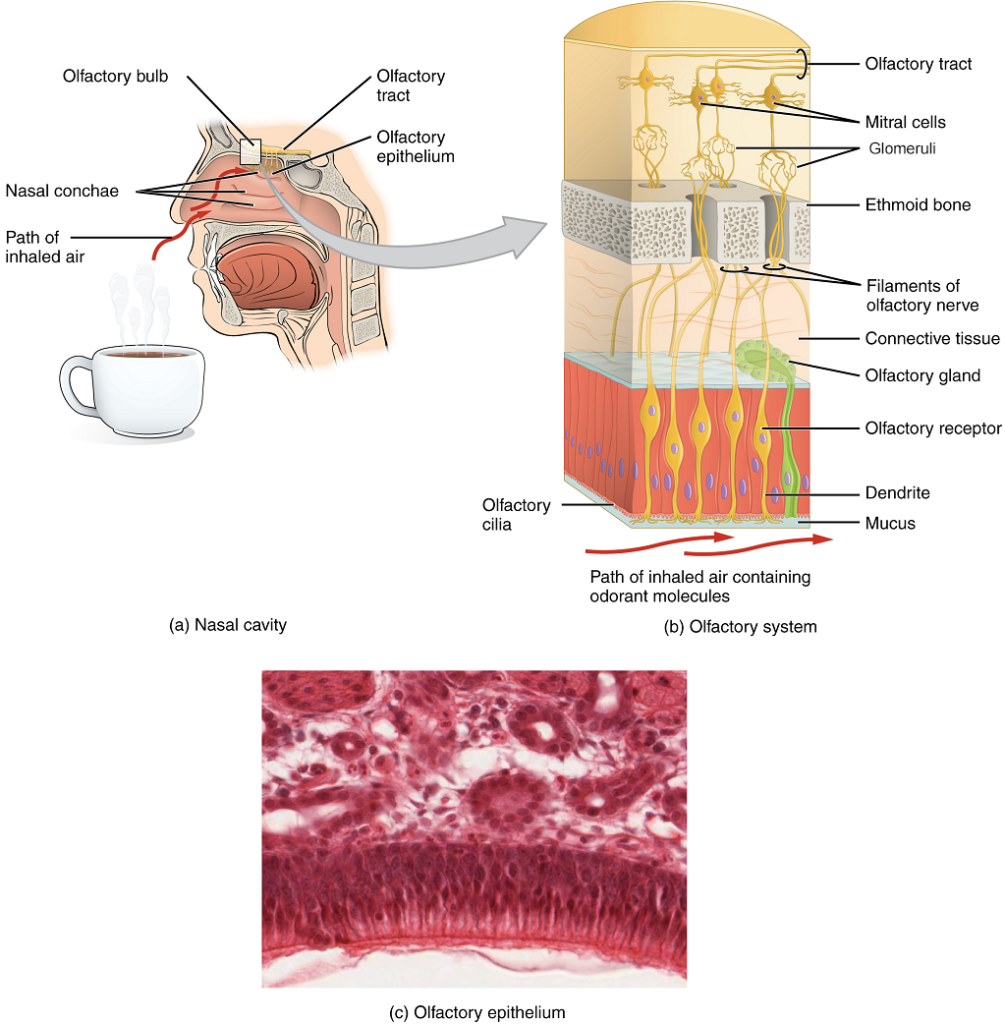
Like taste, the sense of smell, or olfaction, is responsive to chemical stimuli. The olfactory receptor neurons are located in a small region of the walls of the superior nasal cavity (Figure 3). This region is referred to as the olfactory epithelium and contains bipolar sensory neurons. Each olfactory sensory neuron has dendrites that extend from the apical surface of the epithelium into the mucus lining the cavity. As airborne molecules are inhaled through the nose, they pass over the olfactory epithelial region and dissolve into the mucus. These odorant molecules bind to proteins that keep them dissolved in the mucus and help transport them to the olfactory dendrites. The odorant–protein complex binds to a receptor protein within the cell membrane of an olfactory dendrite. These receptors are G protein–coupled, and will produce a graded potential in the olfactory sensory neurons.
The axons of all the olfactory sensory neurons form the olfactory nerve, which extends from the basal surface of the epithelium, through an opening in the skull, and into the brain. The olfactory nerve synapses with neurons in the olfactory bulb on the ventral surface of the frontal lobe. The group of axons called the olfactory tract connect to the olfactory bulb on the ventral surface of the frontal lobe. From there, the axons split to travel to several brain regions. Some travel to the cerebral cortex, specifically to the primary olfactory cortex located in the temporal lobe. Others project to structures within the limbic system and hypothalamus, where smells become associated with long-term memory and emotional responses. This is how certain smells trigger emotional memories, such as the smell of food associated with one’s birthplace. Smell is the one sensory modality which does not synapse in the thalamus before connecting to the cerebral cortex. This intimate connection between the olfactory system and the cerebral cortex is one reason why smell can be a potent trigger of memories and emotion.
The nasal epithelium, including the olfactory neurons, can be harmed by airborne toxic chemicals. Therefore, the olfactory neurons are regularly replaced within the nasal epithelium, and the axons of the new neurons must find their appropriate connections in the olfactory bulb. These new axons grow along the axons that are already in place in the cranial nerve.
Part 5: Audition and Balance
Anatomy of the Ear
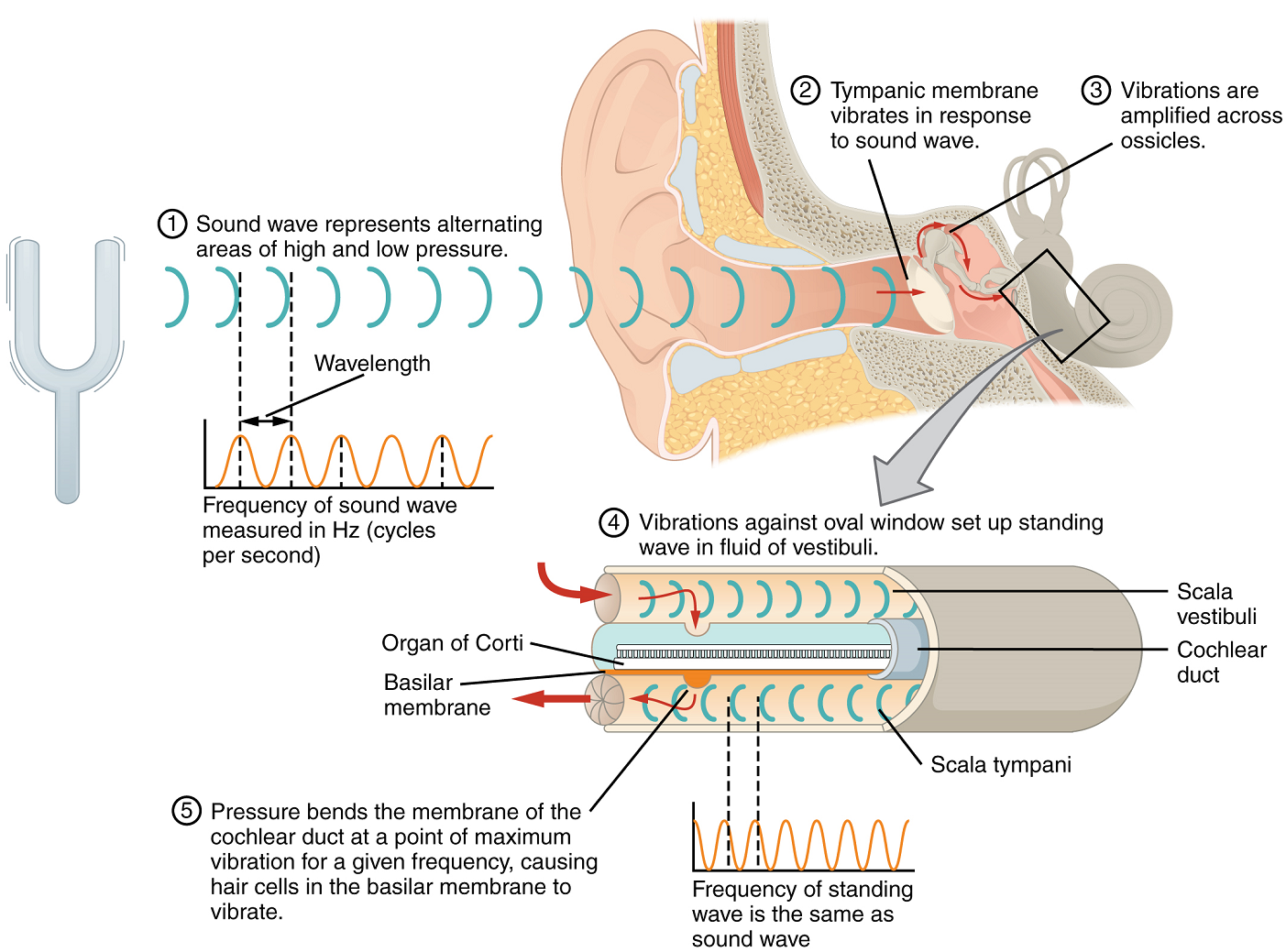
Hearing, or audition, is the transduction of sound waves into a neural signal which is made possible by the structures of the ear (Figure 4). The large, fleshy structure on each lateral aspect of the head is known as the auricle. Some sources also refer to this structure as the pinna, although this term is more appropriate for a structure that can be moved, such as the external ear of a cat. The C-shaped curves of the auricle direct sound waves toward the auditory canal. The canal enters the skull through the external auditory meatus of the temporal bone. At the end of the auditory canal is the tympanic membrane, or eardrum, which vibrates once it is struck by sound waves. The auricle, auditory canal and tympanic membrane are collectively referred to as the external ear. The middle ear consists of a space spanned by three small bones called the auditory ossicles. The three ossicles are the malleus, incus and stapes, which are Latin names that roughly translate into hammer, anvil and stirrup, respectively. The malleus is attached to the tympanic membrane and articulates with the incus. The incus, in turn, articulates with the stapes. The stapes covers an opening (the oval window) leading into the inner ear, where the sound waves will be transduced into a neural signal. The outer and middle ear are responsible for directing sound waves towards the inner ear; interference with this conduction of sound waves through the outer and middle ear can cause conductive deafness if the sound waves fail to reach the inner ear.
The middle ear is connected to the pharynx through the Eustachian tube, which helps equilibrate air pressure across the tympanic membrane. This tube is normally closed but will pop open when the muscles of the pharynx contract during swallowing or yawning. The middle ear is also connected to the mastoid antrum, an air space in the mastoid process of the temporal bone. The middle ear has one more opening covered by a membrane called the round window, which connects with the inner ear.
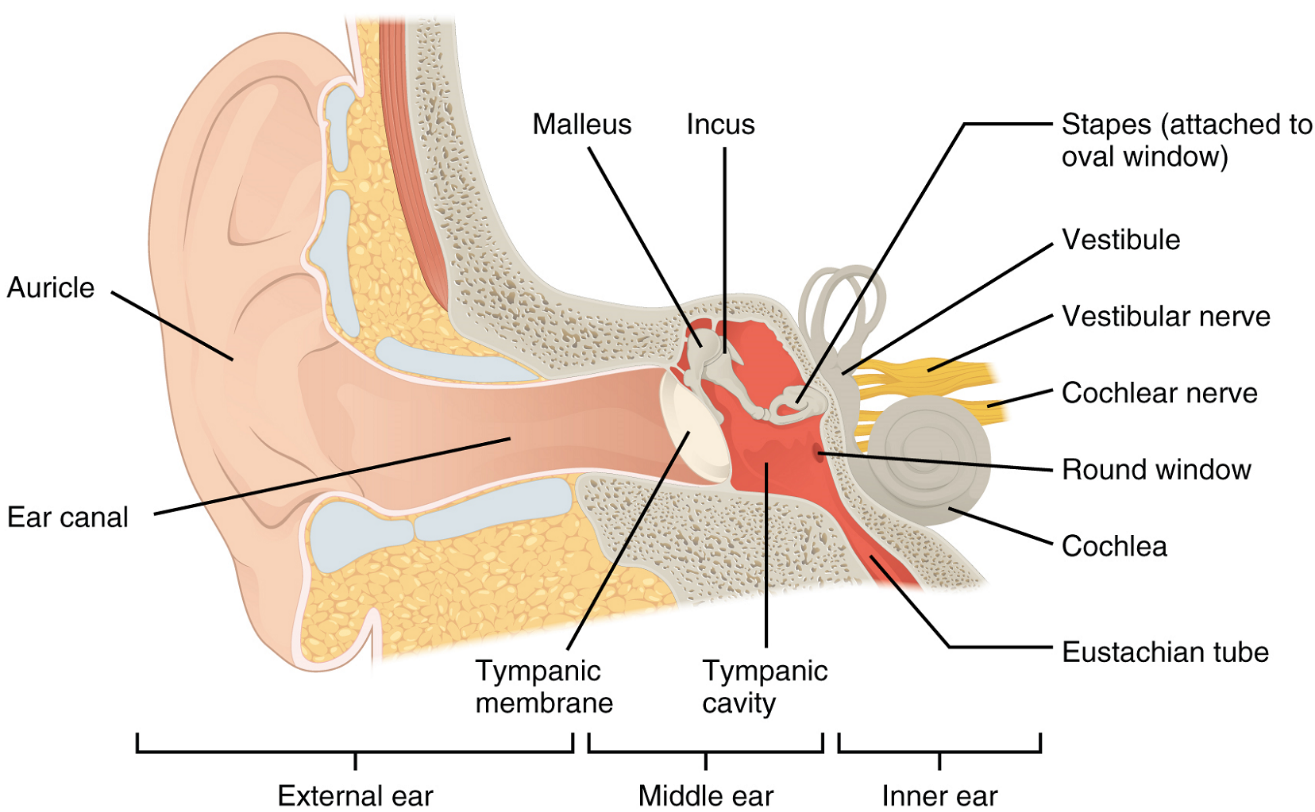
The inner ear is often described as a bony labyrinth, as it is composed of a series of canals embedded within the temporal bone. It has two separate regions, the cochlea and the vestibule, which are responsible for audition (hearing) and equilibrium (balance), respectively. The neural signals from these two regions are relayed to the brainstem through separate fibre bundles. However, these two distinct bundles travel together from the inner ear to the brainstem as the vestibulocochlear nerve. Sound is transduced into neural signals within the cochlear region of the inner ear, which contains the sensory neurons of the spiral ganglia. These ganglia are located within the spiral-shaped cochlea of the inner ear. The cochlea is attached to the stapes through the oval window. Interference in the mechanisms responsible for transducing sound pressure waves to neural signals, or in the passage of information along the cochlear branch of the vestibulocochlear nerve, can result in sensorineural deafness, where a sound wave may reach the inner ear but not ultimately be perceived.
The oval window is located at the beginning of a fluid-filled tube within the cochlea called the scala vestibuli. The fluid within it is perilymph. The scala vestibuli runs along, and above, the cochlear duct (scala media), which is the central cavity of the cochlea containing sound-transducing neurons. At the uppermost tip of the cochlea, the scala vestibuli curves over the top of the cochlear duct. The perilymph-filled tube, now called the scala tympani, returns to the base of the cochlea, this time travelling under the cochlear duct. The scala tympani ends at the round window, covered by a membrane that contains the fluid within the scala. As vibrations of the ossicles travel through the oval window, the fluid of the scala vestibuli and scala tympani moves in a wave-like motion. The frequency of the fluid waves matches the frequencies of the sound waves (Figure 5). The membrane covering the round window will bulge out or pucker in with the movement of the fluid within the scala tympani.
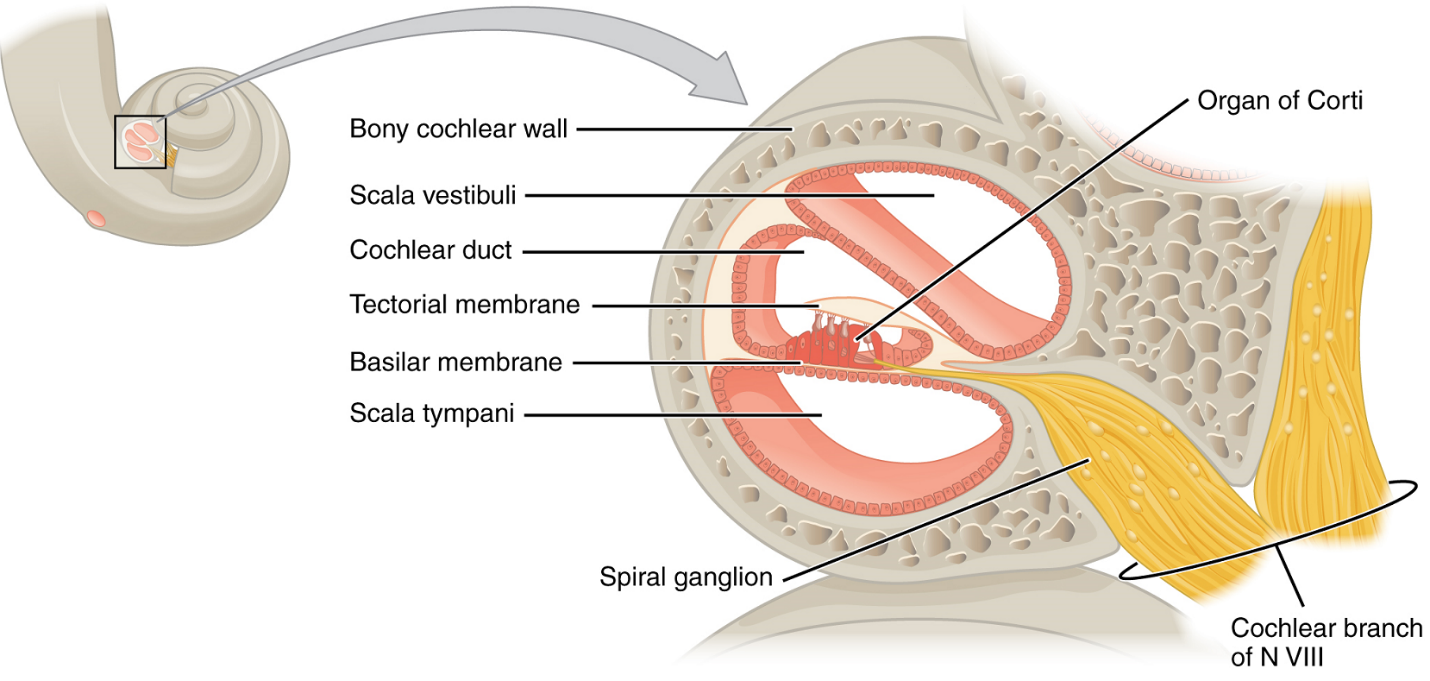
A cross-sectional view of the cochlea shows that the scala vestibuli and scala tympani run along both sides of the cochlear duct (Figure 6). The scala vestibuli is separated from the cochlear duct by the vestibular membrane, and the scala tympani is separated from the cochlear duct by the basilar membrane. The cochlear duct is a fluid-filled cavity containing endolymph. The cochlear duct contains the organ of Corti, which transduces the wave motion of the two scalae into neural signals. The organ of Corti, which is also known as the spiral organ, lies on top of the basilar membrane along the length of the cochlear duct. As the fluid waves move through the scala vestibuli and scala tympani, the basilar membrane moves at a specific spot, depending on the frequency of the waves. Higher frequency waves move a region of the vestibular membrane, then basilar membrane which is close to the base of the cochlea. Lower frequency waves move a region of the vestibular membrane, then basilar membrane which is near the tip of the cochlea.
The organ of Corti contains hair cells, which are named for the hair-like stereocilia extending from the cell’s apical surfaces (Figure 7). There are two main types of hair cells: the inner hair cells and the outer hair cells. Only the inner hair cells serve as hearing receptors. The stereocilia are an array of microvilli-like structures arranged from tallest to shortest. Protein fibers tether adjacent hairs together within each array, such that the array will bend in response to movements of the basilar membrane. The stereocilia extend up from the hair cells to the overlying gel-like tectorial membrane. When the pressure waves of endolymph in the cochlear duct vibrate the basilar membrane, the hair cells move as well. These vibrations bend the stereocilia either toward or away from the tallest member of each array of stereocilia. In inner hair cells, when the stereocilia bend toward the tallest member of their array, tension in the protein tethers opens ion channels in the cell membrane. This will further depolarize the cell membrane, exciting the sensory neurons innervating the hair cells and triggering nerve impulses which travel down the afferent nerve fibers of the cochlear branch of the vestibulocochlear nerve. When the stereocilia bend toward the shortest member of their array, the tension on the tethers slackens and the ion channels close. When no sound is present, and the stereocilia are standing straight, a small amount of tension still exists on the tethers, keeping the membrane potential of the hair cell slightly depolarized. The sterocilia of outer hair cells also bend back and forth in response to the vibrating basilar membrane. However, unlike inner hair cell, they do not serve as sensory receptors, but instead modulate the stiffness of the basilar membrane. This helps inner hair cells be more responsive to certain sounds or protects these cells from potentially damaging sounds.
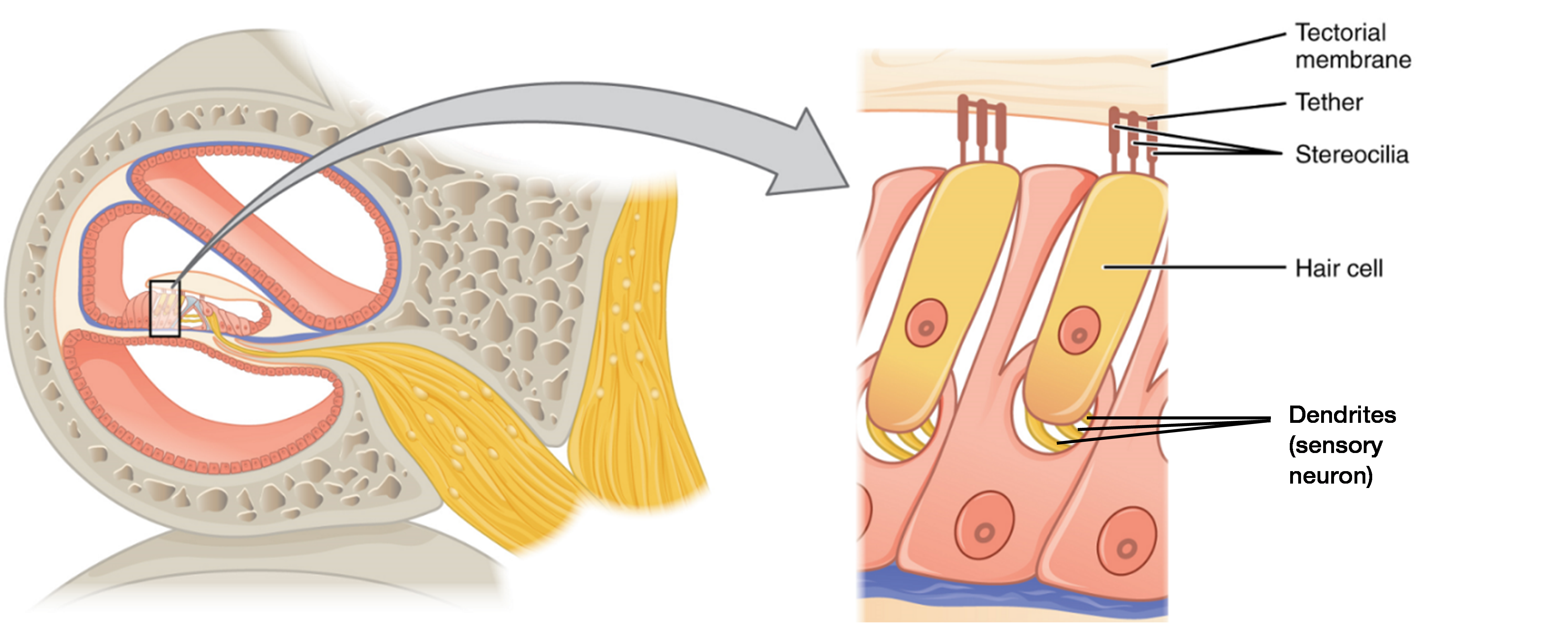
Perilymph, the fluid inside the scala vestibuli, helicotrema and scala tympani, is similar in composition to cerebrospinal fluid. In contrast, endolymph, the fluid in the cochlear duct, contains a relatively high concentration of potassium ions (K+). The depolarization which occurs when stereocilia bend is largely the result of K+ ions (and Ca2+ ions) rushing into the hair cells from the surrounding endolymph through ion channels.
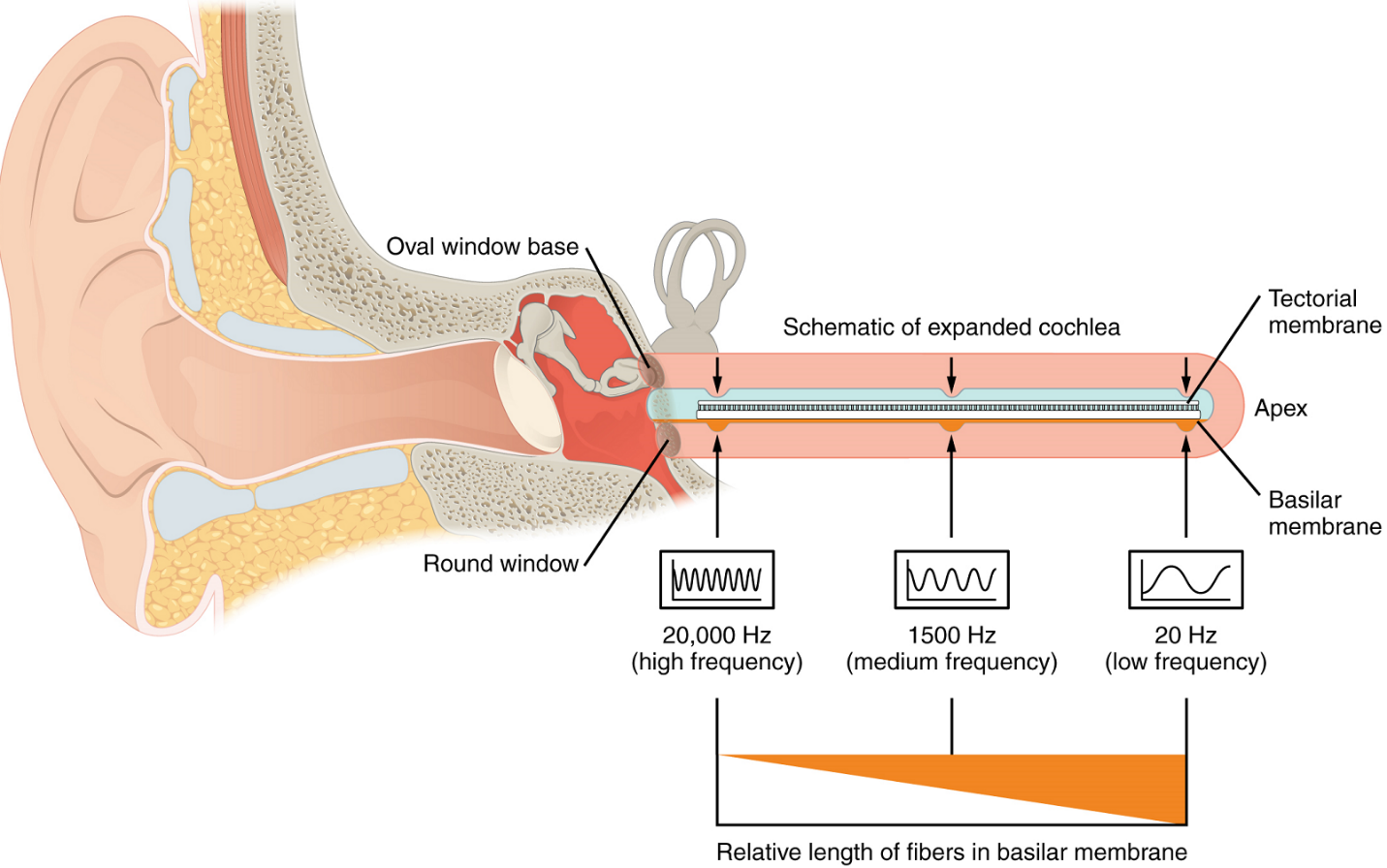
As stated above, a given region of the basilar membrane will only move if the incoming sound is at a specific frequency. Hair cells of the organ of Corti which move in this region will also only respond to sounds of this specific frequency. Therefore, as the frequency of a sound changes, different hair cells of the organ of Corti are activated along the basilar membrane. The cochlea encodes auditory stimuli for frequencies between 20 and 20,000 Hz, which is the range of sound that human ears can detect. The unit of Hertz (Hz) measures the frequency of sound waves in terms of cycles produced per second. Frequencies as low as 20 Hz are detected by hair cells at the wide, floppy apex (tip) of the cochlea. Frequencies in the higher ranges of 20,000 KHz are detected by hair cells at the stiff, narrow base of the cochlea, close to the round and oval windows (Figure 8). Most auditory stimuli contain a mixture of sounds at a variety of frequencies and intensities (represented by the amplitude of the sound wave). The hair cells along the length of the cochlear duct, which are each sensitive to a particular frequency, allow the cochlea to separate auditory stimuli by frequency, just as a prism separates visible light into its component colours. Sounds outside of the ear’s hearing range do reach into the cochlear duct and are thus no perceived by the inner hair cells of the organ of Corti.
Audition: Sensory Pathway
The sensory pathway for audition travels along the vestibulocochlear nerve, which synapses with neurons in the cochlear nuclei of the medulla oblongata. Within the brainstem, input from either ear is combined to extract location information from the auditory stimuli. Whereas the initial auditory stimuli received at the cochlea strictly represent the frequency—or pitch—of the stimuli, the locations of sounds can be determined by comparing information arriving at both ears.
Auditory processing continues on to the midbrain, especially the inferior colliculus. Axons then further project to the thalamus and the superior colliculus. The medial geniculate nucleus of the thalamus receives the auditory information and then projects that information to the primary auditory cortex in the temporal lobe of the cerebral cortex, involved in the conscious awareness of sound. There, the inputs map tonotopically on the cortex – arranged in order of frequency. The superior colliculus receives input from the visual and somatosensory systems, as well as the ears, to initiate stimulation of the muscles that turn the head and neck to orient one’s gaze toward the auditory stimulus.
Balance
Along with audition, the inner ear is responsible for encoding information about equilibrium, the sense of balance (equilibrium). A similar type of mechanoreceptor—a hair cell with stereocilia—senses head position, head movement, and whether our bodies are in motion. These cells are located within the vestibule of the inner ear. Head position and linear acceleration (static equilibrium) is sensed by the utricle and saccule, whereas rotational movement of the head (dynamic equilibrium) is sensed by the semicircular ducts. The neural signals generated in the vestibular ganglion are transmitted through the vestibular branch of the vestibulocochlear nerve to the brainstem and cerebellum.
The utricle and saccule are both largely composed of macula tissue (plural = maculae). The macula is composed of hair cells surrounded by support cells. The stereocilia of the hair cells extend into a viscous gel called the otolithic membrane (Figure 9). On top of the otolithic membrane is a layer of calcium carbonate crystals, called otoliths. The otoliths essentially make the otolithic membrane top-heavy. The otolithic membrane moves separately from the macula in response to head movements. Tilting the head causes the otolithic membrane to slide over the macula in the direction of gravity. The moving otolithic membrane, in turn, bends the sterocilia, causing some hair cells to depolarize, as others hyperpolarize. The exact position of the head is interpreted by the brain based on the pattern of hair-cell depolarization.
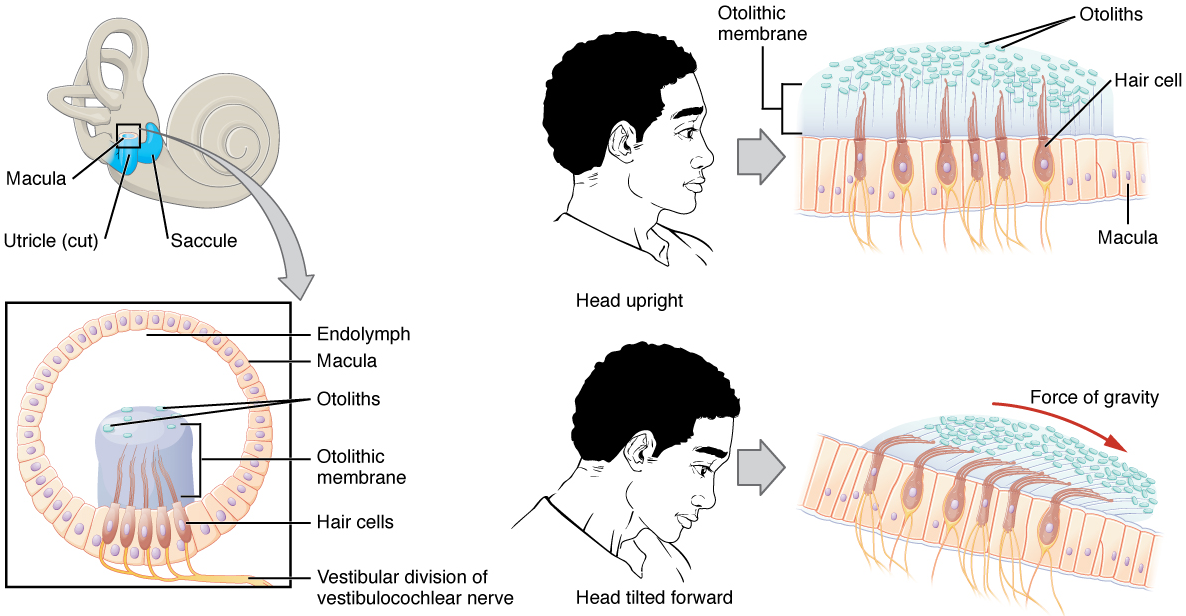
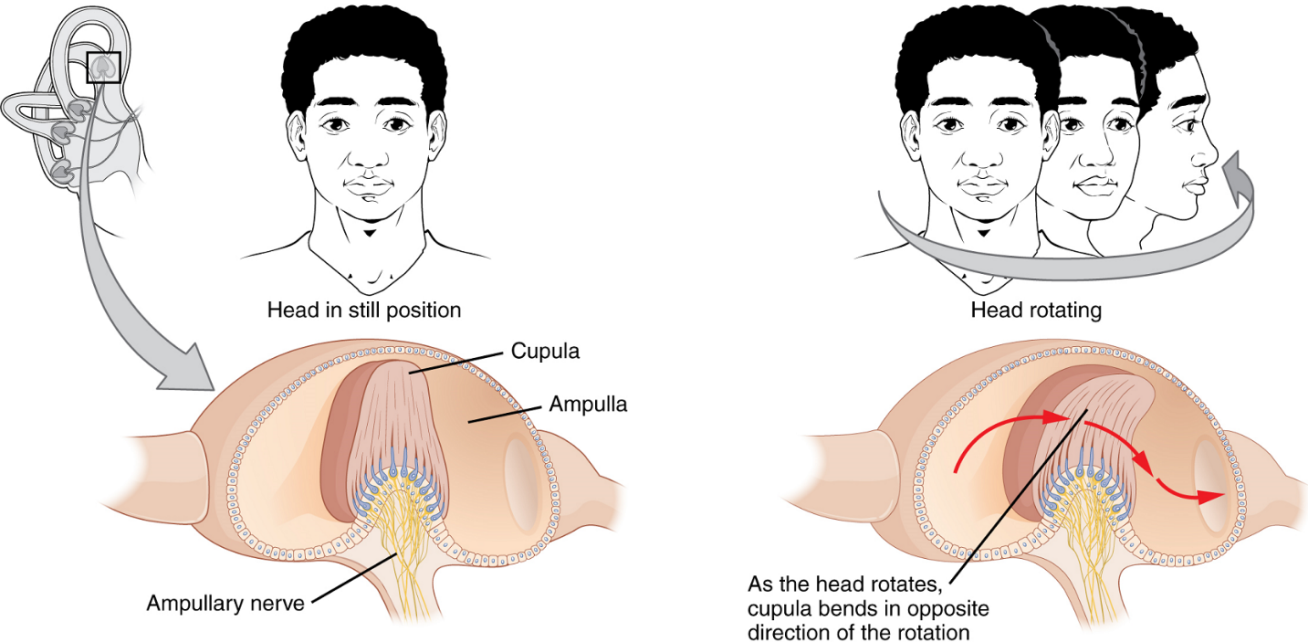
The semicircular canals are three ring-like extensions of the vestibule. One is oriented in the horizontal plane, whereas the other two are oriented in the vertical plane. The anterior and posterior vertical canals are oriented at approximately 45 degrees relative to the sagittal plane (Figure 10). Within each canal is an additional compartment called the semicircular duct. While the semicircular canals are filled with perilymph, the semicircular ducts within are filled with endolymph. The base of each semicircular canal is an enlarged region known as the ampulla. Each ampulla contains a sense organ of balance named crista ampullaris, which responds to rotational movement, such as turning the head while saying “no.” This sense organ contains hair cells, with stereocilia on the apical side. The stereocilia extend into the cupula, a jelly-like structure which attaches to the top of the ampulla. As the head rotates in a plane parallel to the semicircular duct, the fluid lags, deflecting the cupula in the direction opposite to the head movement. Some ampullae of the canals are oriented horizontally and others are oriented vertically. By comparing the relative movements of both the horizontal and vertical ampullae, the vestibular system can detect the direction of most head movements within three-dimensional (3-D) space.
Balance is coordinated through the vestibular system, the nerves of which are composed of axons from the vestibular ganglion that carries information from the utricle, saccule and semicircular ducts. The system contributes to controlling head and neck movements in response to vestibular signals. An important function of the vestibular system is coordinating eye and head movements to maintain visual attention. Most of the axons terminate in the vestibular nuclei of the medulla oblongata.
Some axons project from the vestibular ganglion directly to the cerebellum, with no intervening synapse in the vestibular nuclei. The cerebellum is primarily responsible for initiating movements on the basis of equilibrium information.
Neurons in the vestibular nuclei project their axons to targets in the brainstem. One target is the reticular formation, which influences respiratory and cardiovascular functions in relation to body movements. A second target of the axons of neurons in the vestibular nuclei is the spinal cord, which initiates the spinal reflexes involved with posture and balance. To assist the visual system, fibers of the vestibular nuclei project to the oculomotor, trochlear and abducens nuclei to influence signals sent along the cranial nerves. Finally, the vestibular nuclei project to the thalamus to join the proprioceptive pathway, allowing conscious perception of equilibrium.
Part 6: Vision
Anatomy of the Eye
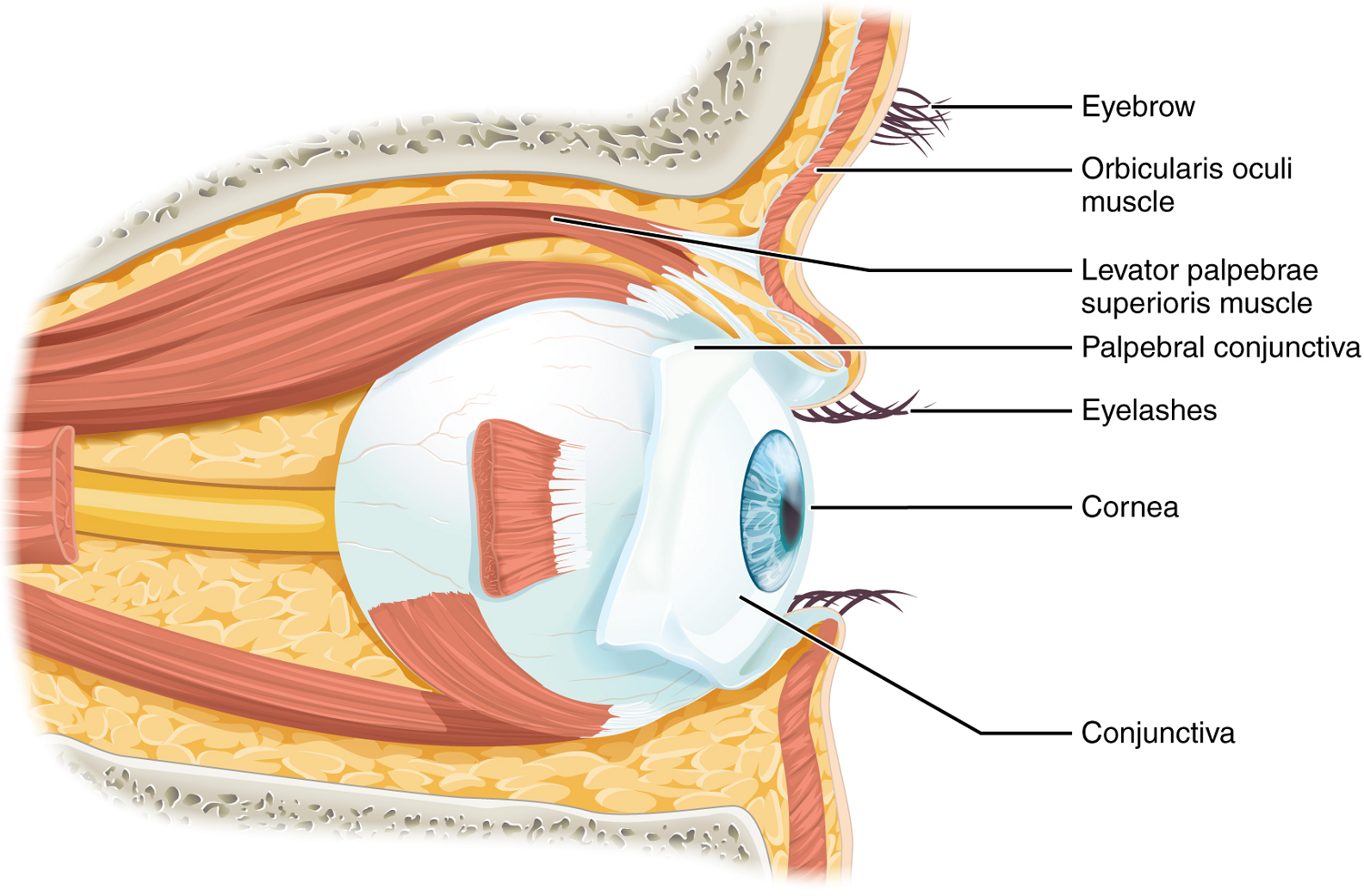
Vision is the special sense of sight that is based on the transduction of light stimuli received through the eyes. The eyes are located within either orbit in the skull. The bony orbits surround the eyeballs, protecting them and anchoring the soft tissues of the eye (Figure 11). The eyelids, with lashes at their leading edges, help to protect the eye from abrasions by blocking particles that may land on the surface of the eye. The inner surface of each lid is a thin membrane known as the palpebral conjunctiva. The conjunctiva extends over the white areas of the eye (the sclera), connecting the eyelids to the eyeball. Tears are produced by the lacrimal gland, located beneath the lateral edges of the nose on each side of the nose. Tears produced by this gland flow through the lacrimal duct to the medial corner of the eye, where the tears flow over the conjunctiva, washing away foreign particles.
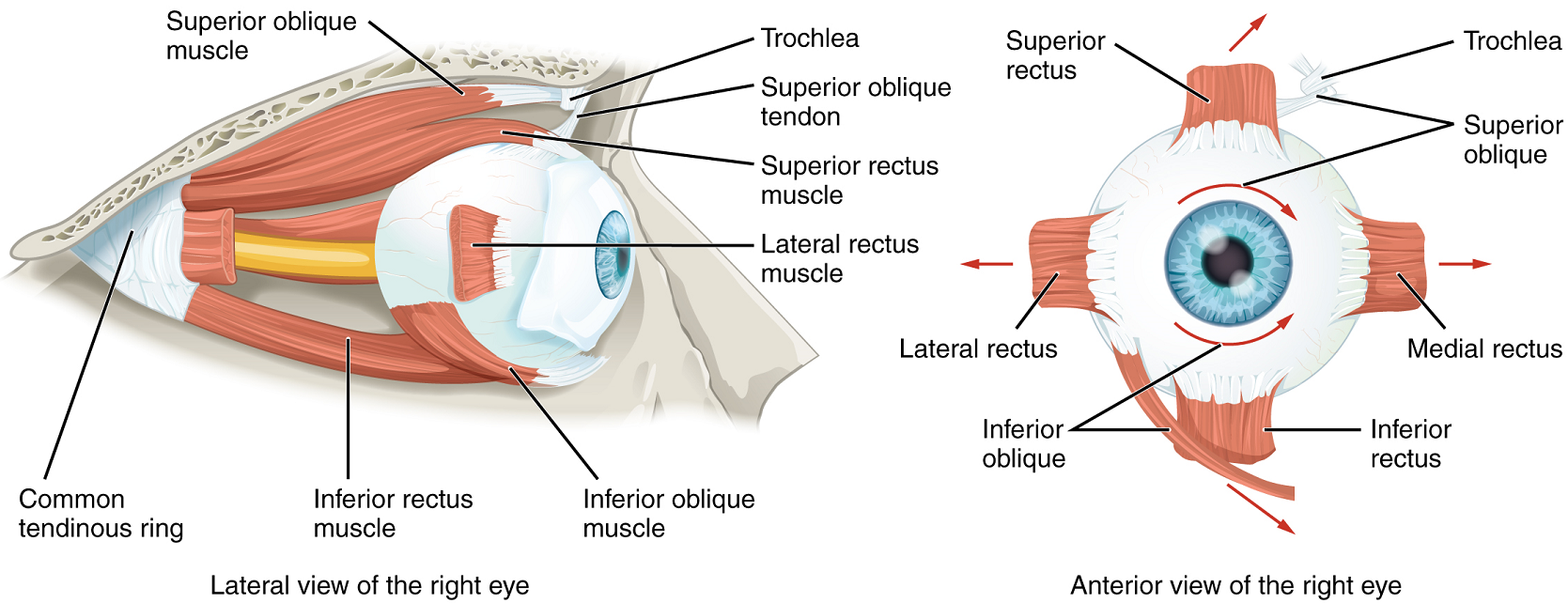
Movement of the eye within the orbit is accomplished by the contraction of six extraocular muscles which originate from the bones of the orbit and insert into the surface of the eyeball (Figure 12).
The eye itself is a hollow sphere composed of three layers of tissue (tunics). The outermost layer is the fibrous tunic, which includes the white sclera and clear cornea. The sclera accounts for five sixths of the surface of the eye, most of which is not visible, though humans are unique compared with many other species in having so much of the “white of the eye” visible (Figure 13). The transparent cornea covers the anterior tip of the eye and allows light to enter the eye. The middle layer of the eye is the vascular tunic, which is mostly composed of the choroid, ciliary body and iris. The choroid is a layer of highly vascularized connective tissue that provides a blood supply to the eyeball. The choroid is posterior to the ciliary body, a muscular structure that is attached to the lens by zonule fibres (suspensory ligaments). These two structures bend the lens, allowing it to focus light on the back of the eye.
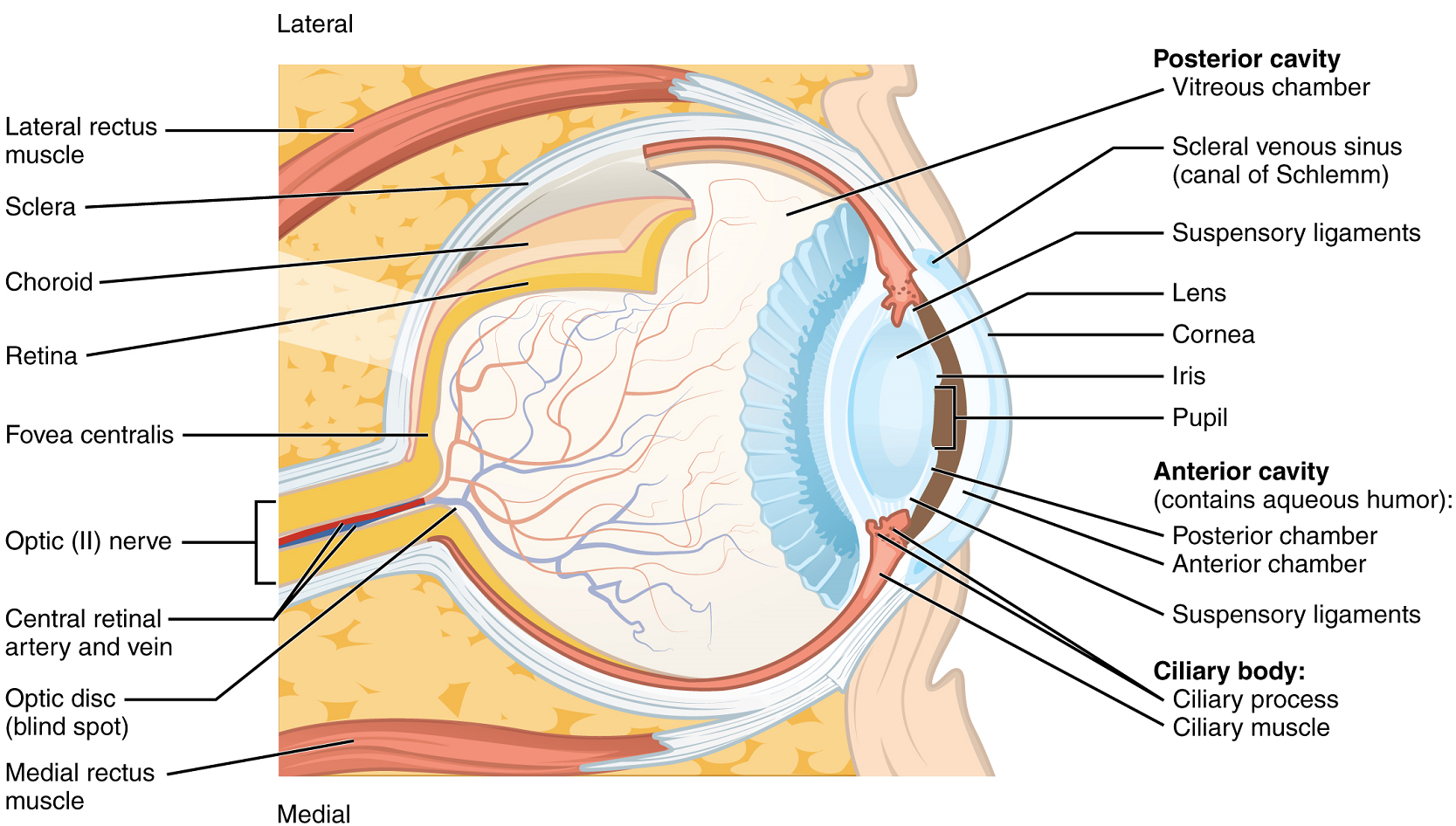
Overlaying the ciliary body, and visible in the anterior eye, is the iris—the colored part of the eye. The iris is composed of two smooth muscles –the circular sphincter pupillae and the radial dilator pupillae which open and close the pupil, the hole at the centre of the eye that allows light to enter. The sphincter pupillae contracts in response to parasympathetic nervous system activation, contracting the pupil, whereas the dilator pupillae contracts in response to sympathetic nervous system activation, dilating the pupil. The iris generally constricts the pupil in response to bright light and dilates the pupil in response to dim light, to regulate the amount of light reaching the innermost layer of the eye. This innermost layer is the neural tunic, or retina, which contains the nervous tissue responsible for photoreception.
The eye is also divided into two cavities: the anterior cavity (chamber) and the posterior cavity (chamber). The anterior cavity is the space between the cornea and lens, including the iris and ciliary body. It is filled with a watery fluid called the aqueous humour. The posterior cavity is the space behind the lens that extends to the posterior side of the interior eyeball, where the retina is located. The posterior cavity is filled with a more viscous fluid called the vitreous humour.
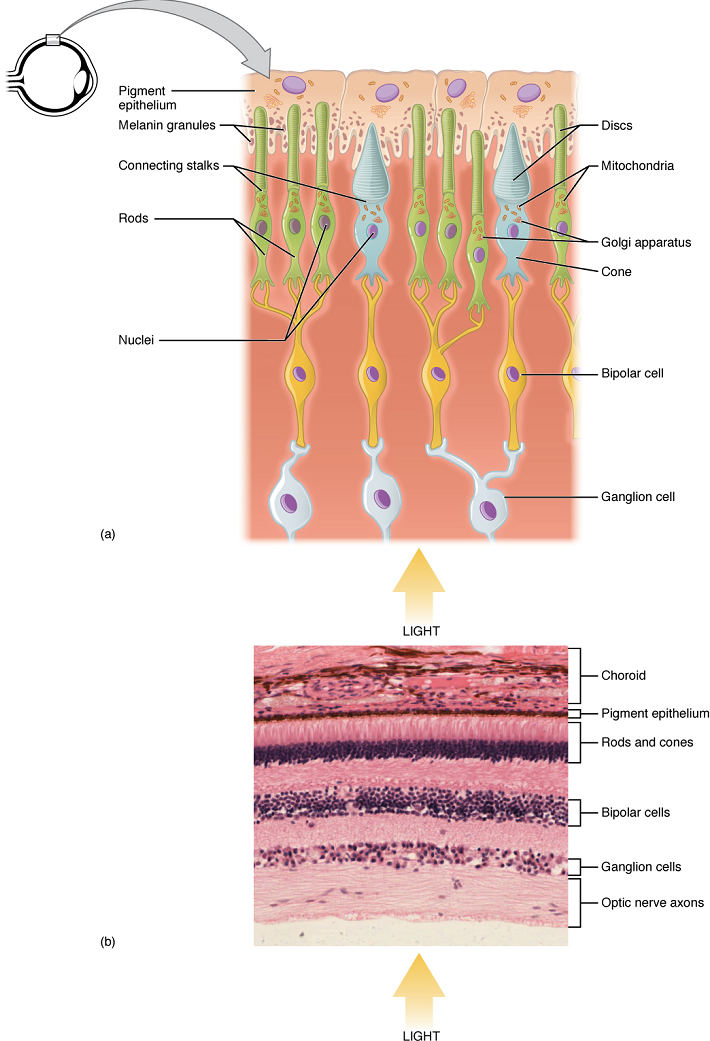
The retina is composed of several layers and contains specialized cells for the initial processing of visual stimuli. The photoreceptors (rods and cones) change their membrane potential when stimulated by light energy. The change in membrane potential alters the amount of neurotransmitter that the photoreceptor cells release onto bipolar cells in the outer synaptic layer. It is the bipolar cell in the retina that connects a photoreceptor to a ganglion cell in the inner synaptic layer. There, amacrine cells additionally contribute to retinal processing before an action potential is produced by the ganglion cells. The axons of ganglion cells, which lie in the innermost layer of the retina, collect at the optic disc and leave the eye as the optic nerve (see Figure 13). Because these axons pass through the retina, there are no photoreceptors at the very back of the eye, where the optic nerve begins. This creates a “blind spot” in the retina, and a corresponding blind spot in our visual field.
Note that the photoreceptors in the retina (rods and cones) are located behind the axons, ganglion cells, bipolar cells and retinal blood vessels. A significant amount of light is absorbed by these structures before the light reaches the photoreceptor cells. However, at the exact centre of the retina is a small area known as the macula lutea with a small depression in the middle called the fovea. At the fovea, the retina lacks the supporting cells and blood vessels, and only contains photoreceptors. The fovea is where the least amount of incoming light is absorbed by other retinal structures (see Figure 13). Therefore, visual acuity, or the sharpness of vision, is greatest at the fovea. As one moves in either direction from this central point of the retina, visual acuity drops significantly. In addition, each photoreceptor cell of the fovea is connected to a single ganglion cell. Therefore, this ganglion cell does not have to integrate inputs from multiple photoreceptors, which reduces the accuracy of visual transduction. Toward the edges of the retina, several photoreceptors converge on ganglion cells (through the bipolar cells) up to a ratio of 50 to 1. The difference in visual acuity between the fovea and peripheral retina is easily evidenced by looking directly at a word in the middle of this paragraph. The visual stimulus in the middle of the field of view falls on the fovea and is in the sharpest focus. Without moving your eyes off that word, notice that words at the beginning or end of the paragraph are not in focus. The images in your peripheral vision are focused on the peripheral retina, and have vague, blurry edges and words that are not as clearly identified. As a result, a large part of the neural function of the eyes is concerned with moving the eyes and head so that important visual stimuli are centered on the fovea. Finally, there is a high concentration of cones (rather than rods) in the fovea, which allow the detection of different colours as described below.
Photoreceptors
Light falling on the retina causes chemical changes to pigment molecules in the photoreceptors, ultimately leading to a change in the activity of the ganglion cells. Photoreceptor cells have two parts, the inner segment and the outer segment (Figure 14). The inner segment contains the nucleus and other common organelles of a cell, whereas the outer segment is a specialized region in which photoreception takes place. There are two types of photoreceptors—rods and cones—which differ in the shape of their outer segment.
The rod-shaped outer segments of the rod photoreceptor contain a stack of membrane-bound discs that contain the photosensitive pigment rhodopsin. The cone-shaped outer segments of the cone photoreceptor contain their photosensitive pigments in infoldings of the cell membrane. There are three cone photopigments, called opsins, which are each sensitive to a particular wavelength of light. The wavelength of visible light determines its colour. The pigments in human eyes are specialized in perceiving three different primary colours: red, green and blue.
At the molecular level, visual stimuli cause changes in the photopigment molecule that lead to changes in the membrane potential of the photoreceptor cell. A single unit of light is called a photon, which is described in physics as a packet of energy with properties of both a particle and a wave. The energy of a photon is represented by its wavelength, with each wavelength of visible light corresponding to a particular colour. Visible light is electromagnetic radiation with a wavelength between 380 and 720 nm. Wavelengths of electromagnetic radiation longer than 720 nm fall into the infrared range, whereas wavelengths shorter than 380 nm fall into the ultraviolet range. Light with a wavelength of 380 nm is blue whereas light with a wavelength of 720 nm is dark red. All other colours fall between red and blue at various points along the wavelength scale.
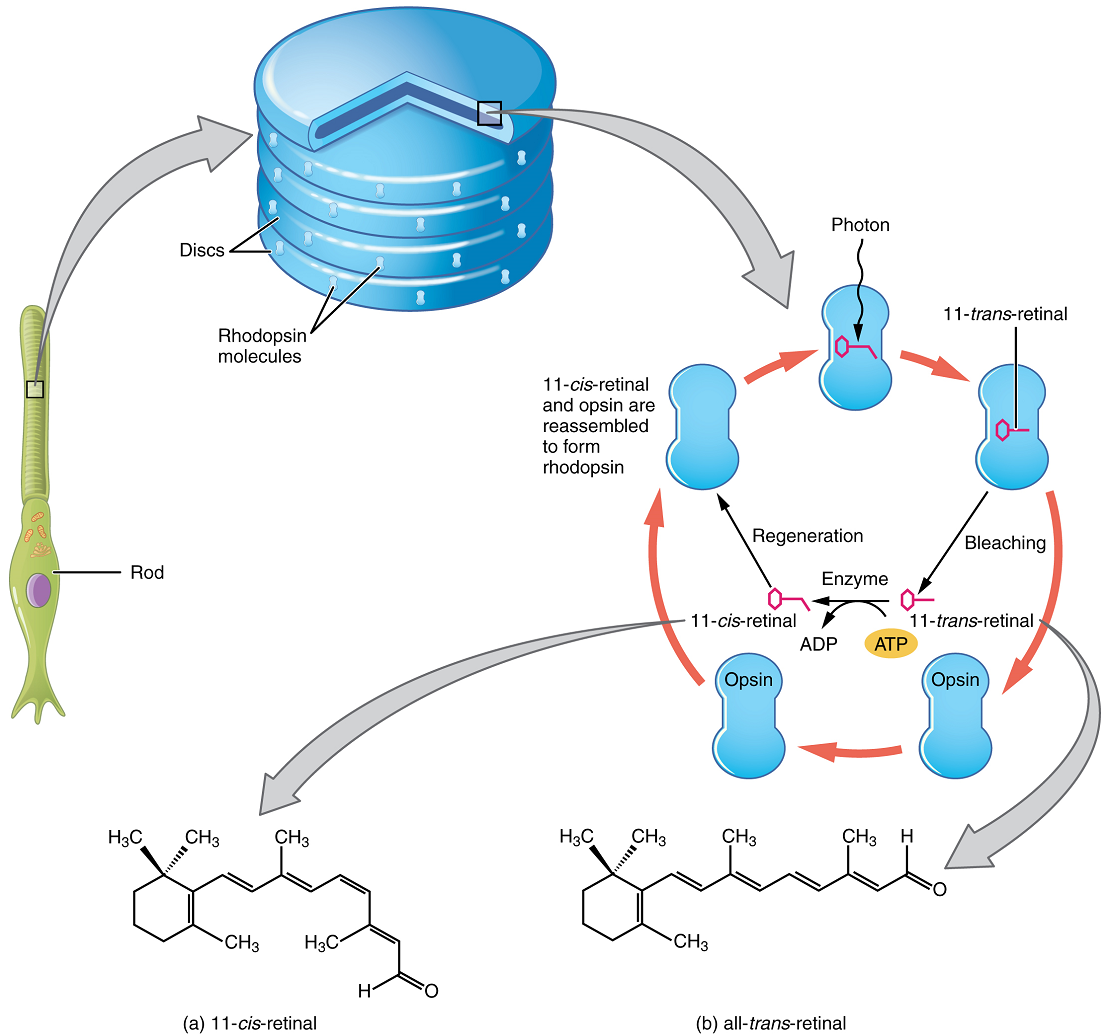
Opsin pigments are actually transmembrane proteins that contain a cofactor known as retinal. Retinal is a hydrocarbon molecule related to vitamin A. When a photon hits retinal, the long hydrocarbon chain of the molecule is biochemically altered. This process is called photoisomerization. Before interacting with a photon, retinal’s flexible double-bonded carbons are in the cis conformation. This molecule is referred to as 11-cis-retinal. A photon interacting with the molecule causes the flexible double-bonded carbons to change to the trans conformation, forming all-trans-retinal, which has a straight hydrocarbon chain (Figure 15).
The shape change of retinal in the photoreceptors initiates visual transduction in the retina. Activation of retinal and the opsin proteins result in the activation of a G protein. The G protein changes the membrane potential of the photoreceptor cell, causing a slight hyperpolarization, and the cell then releases less neurotransmitter into the outer synaptic layer of the retina. Until the retinal molecule is changed back to the 11-cis-retinal shape, the opsin cannot respond to light energy, a process called bleaching. When a large group of photopigments is bleached, the retina will send information as if opposing visual information is being perceived. After a bright flash of light, the afterimages are usually seen in negative. The photoisomerization is reversed by a series of enzymatic changes so that the retinal responds to more light energy.
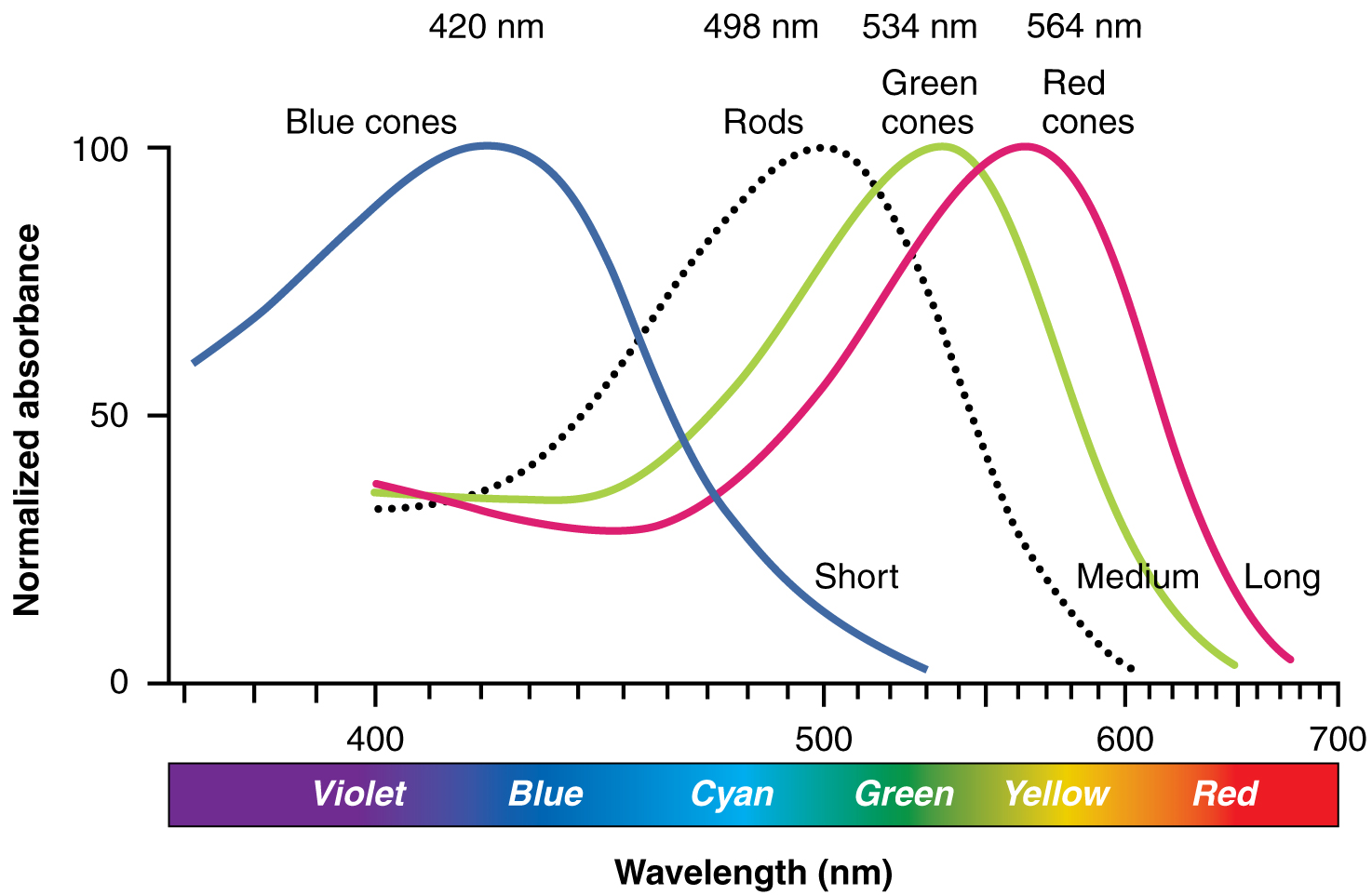
The opsins are sensitive to limited wavelengths of light. Rhodopsin, the photopigment in rods, is most sensitive to light at a wavelength of 498 nm. The three colour opsins have peak sensitivities of 564 nm, 534 nm and 420 nm, corresponding roughly to the primary colours of red, green and blue (Figure 16). The absorbance of rhodopsin in the rods is much more sensitive than in the cone opsins; specifically, rods are sensitive enough to be excited in low-light conditions, whereas cones are sensitive to brighter conditions. In normal sunlight, rhodopsin will be constantly bleached while the cones are active. In a darkened room, there is not enough light to activate cone opsins, and vision is entirely dependent on rods. Rods are so sensitive to light that a single photon can result in an action potential from a rod’s corresponding ganglion cell.
The three types of cone opsins, being sensitive to different wavelengths of light, provide us with colour vision. By comparing the activity of the three different types of cones, the brain can extract colour information from visual stimuli. For example, a bright blue light that has a wavelength of approximately 450 nm would activate the “red” cones minimally, the “green” cones marginally, and the “blue” cones predominantly. The relative activation of the three different cones is calculated by the brain, which perceives the colour as blue. However, cones cannot react to low-intensity light, and rods do not sense the colour of light. Therefore, our low-light vision is—in essence—in grayscale. In other words, in a dark room, everything appears as a shade of gray. If you think that you can see colours in the dark, it is most likely because your brain knows what colour something is and is relying on that memory.
Focusing Light on the Retina
To see an object in sharp focus and in colour, the light rays from that object must travel to the fovea of the retina. This is largely accomplished through a combination of contractions or relaxations of the appropriate extraocular muscles, as well as the accommodation of the lens.
The extraocular muscles allow for greater or lesser convergence of the eyeballs, so that both eyeballs can be directed at the same point in space. When looking at a distant object, both eyes are pointed roughly parallel to each other. As the eyeballs lie at a fixed distance from each other, when focusing on a nearby object the eyes must rotate medially to direct both eyes towards the object being viewed. The closer the object, the greater the degree of convergence required.
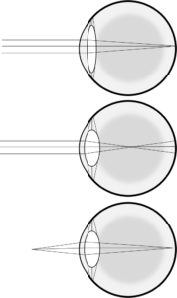
When light rays pass from one medium (e.g. air) to another (e.g. water), they tend to bend, or “refract”. This refraction of light allows light rays entering the eye to be pulled closer together to meet at the fovea. The cornea is responsible for the majority of refraction occurring as light rays enter the eyeball, but the cornea itself is not adjustable, so this refraction is constant and cannot be used to focus on objects at different distances. However, additional refraction occurs as the light passes through the lens, and the shape and thickness of the lens can be modified to control the degree of refraction (Figure 17). This change in shape of the lens to produce more or less refraction of incoming light rays is known as accommodation, and is accomplished by the contraction or relaxation of the circular ciliary muscle in the ciliary body to which the suspensory ligaments are attached. When the ciliary muscle contracts, it allows the suspensory ligaments to loosen and the lens to bulge. When the ciliary muscle relaxes, it pulls the suspensory ligaments taut, pulling the lens flat.
In addition to their role regulating the total amount of light striking the retina, the pupillary muscles also participate in allowing focused vision by limiting the amount of light hitting the edges of the lens specifically. When viewing a distant object, the lens is relatively flat, so there are relatively equivalent changes in light direction at the edges relative to the centre. However, when viewing a nearby object the lens bulges and light entering near the edges could result in a distorted or blurry image as a result of spherical aberration. Constricting the pupil by contracting the sphincter pupillae muscle and relaxing the dilator pupillae muscle covers the edge of the lens with the iris, eliminating this distortion. When viewing a distant object the sphincter pupillae muscle tends to relax and the dilator pupillae contracts, thus dilating the pupil.
The Visual Pathway
Once any sensory cell transduces a stimulus into a nerve impulse, the impulse has to travel along axons to reach the CNS. In many of the special senses, the axons leaving the sensory receptors have a topographical arrangement, meaning that the location of the sensory receptor relates to the location of the axon in the nerve. For example, in the retina, axons from ganglion cells in the fovea are located at the centre of the optic nerve, where they are surrounded by axons from the more peripheral ganglion cells.
The axons from the medial retina of the left eye cross over to the right side of the brain at the optic chiasm. However, within each eye, the axons projecting from the lateral side of the retina do not decussate. For example, the axons from the lateral retina of the right eye project back to the right side of the brain. Therefore, the left field of view of each eye is processed on the right side of the brain, whereas the right field of view of each eye is processed on the left side of the brain (Figure 18).
A unique clinical presentation that relates to this anatomic arrangement is the loss of lateral peripheral vision, known as bilateral hemianopia. This is different from “tunnel vision” because the superior and inferior peripheral fields are not lost. Visual field deficits can be disturbing for a patient, but in this case, the cause is not within the visual system itself. A growth of the pituitary gland presses against the optic chiasm and interferes with signal transmission. However, the axons projecting to the same side of the brain are unaffected. Therefore, the patient loses the outermost areas of their field of vision and cannot see objects to their right and left.
Extending from the optic chiasm, the axons of the visual system are referred to as the optic tract instead of the optic nerve. The optic tract has three major targets, two in the diencephalon and one in the midbrain. The majority of the connections of the optic tract are to the thalamus—specifically, the lateral geniculate nucleus. Axons from this nucleus then project to the visual cortex of the cerebrum, located in the occipital lobe. Another target of the optic tract are the superior colliculi, which are visual reflex centres in the midbrain.
In addition, a very small number of ganglion cell axons project from the optic chiasm to the hypothalamus. These ganglion cells are photosensitive, in that they respond to the presence or absence of light. Unlike photoreceptors, however, these photosensitive ganglion cells cannot be used to perceive images. By simply responding to the absence or presence of light, these ganglion cells can send information about day length. The perceived proportion of sunlight to darkness establishes the circadian rhythm of our bodies, allowing certain physiological events to occur at approximately the same time every day.
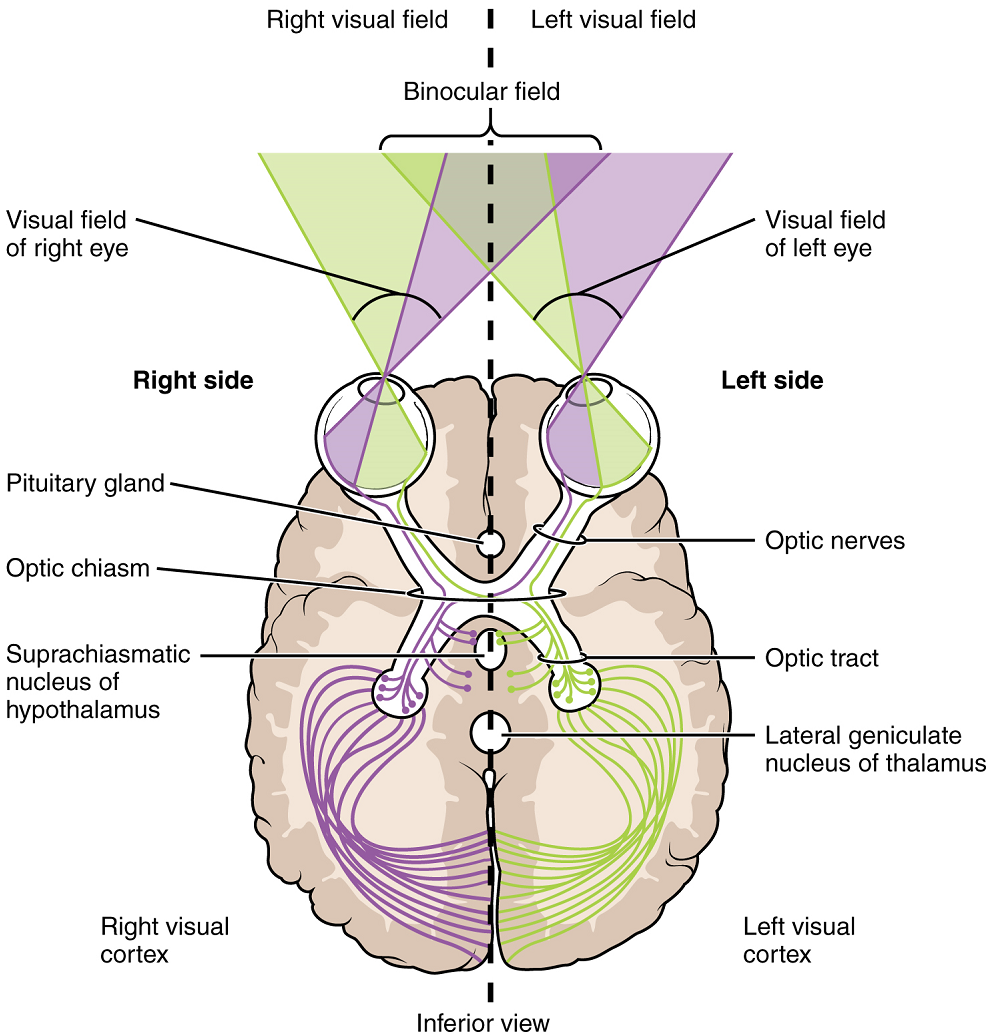
The topographic relationship between the retina and the visual cortex is maintained throughout the visual pathway. The visual field is projected onto the two retinae, as described above, with sorting at the optic chiasm. The right peripheral visual field falls on the medial portion of the right retina and the lateral portion of the left retina. The right medial retina then projects across the midline through the optic chiasm. This results in the right visual field being processed in the left visual cortex. Likewise, the left visual field is processed in the right visual cortex (Figure 18). Though the chiasm is helping to sort right and left visual information, superior and inferior visual information is maintained topographically in the visual pathway. Light from the superior visual field falls on the inferior retina, and light from the inferior visual field falls on the superior retina. This topography is maintained such that the superior region of the visual cortex processes the inferior visual field and vice versa. Therefore, the visual field information is inverted and reversed as it enters the visual cortex—up is down, and left is right. However, the cortex processes the visual information such that the final conscious perception of the visual field is correct. The topographic relationship is evident in that information from the foveal region of the retina is processed in the centre of the primary visual cortex. Information from the peripheral regions of the retina are correspondingly processed toward the edges of the visual cortex.
Part 1: Overall Organization of the Sensory Systems
Part 2: Gustation
Part 3: Olfaction
Part 4: Audition and Balance
Part 5: Vision
Cell that transduces environmental stimuli into neural signals.
Process of changing an environmental stimulus into the electrochemical signals of the nervous system.
Configuration of a sensory receptor neuron with dendrites in the connective tissue of the organ, such as in the dermis of the skin, that are most often sensitive to chemical, thermal, and mechanical stimuli.
Configuration of a sensory receptor neuron with dendrites surrounded by specialized structures to aid in transduction of a particular type of sensation, such as the lamellated corpuscles in the deep dermis and subcutaneous tissue.
Cell that transduces environmental stimuli into neural signals.
Encapsulated mechanoreceptor cell found in the skin that responds to pressure and touch.
Receptor cell specialized to respond to light stimuli.
Sensory receptor that is positioned to interpret stimuli from the external environment, such as photoreceptors in the eye or somatosensory receptors in the skin.
Sensory receptor that is positioned to interpret stimuli from internal organs, such as stretch receptors in the wall of blood vessels.
Largest artery in the body, originating from the left ventricle and descending to the abdominal region where it bifurcates into the common iliac arteries at the level of the fourth lumbar vertebra; arteries originating from the aorta distribute blood to virtually all tissues of the body.
Small pocket near the base of the internal carotid arteries that are the locations of the baroreceptors and chemoreceptors that trigger a reflex that aids in the regulation of vascular homeostasis.
Receptor cell that senses changes in the position and kinesthetic aspects of the body.
Sensory receptor cell that is sensitive to chemical stimuli, such as in taste, smell, or pain.
Receptor cell that senses differences in the concentrations of bodily fluids on the basis of osmotic pressure.
Receptor cell that senses pain stimuli.
Receptor cell that transduces mechanical stimuli into an electrochemical signal.
Sense of position and movement of the body.
Sense of body movement based on sensations in skeletal muscles, tendons, joints, and the skin.
Any sensory system associated with a specific organ structure, namely smell, taste, sight, hearing, and balance.
A particular system for interpreting and perceiving environmental stimuli by the nervous system.
Sensing pain.
Receptor cell that senses pain stimuli.
Sense of touch.
Sense of position and movement of the body.
Sensation of stimuli from internal organs.
Sense of body movement based on sensations in skeletal muscles, tendons, joints, and the skin.
Skin.
Receptor cell that senses temperature stimuli.
Receptor cell in the stratum basale of the epidermis that responds to the sense of touch.
Deepest layer of the epidermis, made of epidermal stem cells.
Encapsulated mechanoreceptor cell found in the skin that responds to pressure and touch.
Layer of skin between the epidermis and hypodermis, composed mainly of connective tissue and containing blood vessels, hair follicles, sweat glands, and other structures.
Cavity or sac from which hair originates.
(Also, Ruffini corpuscle) Cutaneous receptors sensitive to skin stretch and sustained pressure.
Receptor cell that senses changes in the position and kinesthetic aspects of the body.
For gustation (taste), a bump-like projection on the surface of the tongue that contains taste buds.
Structures within a papilla on the tongue that contain gustatory receptor cells.
Sensory cells in the taste bud that transduce the chemical stimuli of gustation.
Change in a cell membrane potential from rest toward or above zero (as during an action potential).
Change in the membrane potential that varies in size, depending on the size of the stimulus that elicits it.
Ninth cranial nerve; responsible for contraction of muscles in the tongue and throat and for part of the sense of taste, as well as causing saliva production.
Tenth cranial nerve; responsible for the autonomic control of organs in the thoracic and upper abdominal cavities.
(In nervous system) a localized collection of neuron cell bodies that are functionally related; a “center” of neural function (plural= nuclei).
Lowest (most inferior) part of the brain, controlling many autonomic functions including heart rate, breathing, and digestion.
Region of the nasal epithelium where olfactory neurons are located.
Receptor cell of the olfactory system, sensitive to the chemical stimuli of smell, the axons of which compose the first cranial nerve.
Type of tissue that serves primarily as a covering or lining of body parts, protecting the body; it also functions in absorption, transport, and secretion.
Single process of the neuron that carries an electrical signal (action potential) away from the cell body toward a target cell.
Central target of the first cranial nerve; located on the ventral surface of the frontal lobe in the cerebrum.
Outer gray matter covering the forebrain, marked by wrinkles and folds known as gyri and sulci.
Structures at the edge (limit) of the boundary between the forebrain and hindbrain that are most associated with emotional behavior and memory formation.
Narrow junction across which a chemical signal passes from neuron to the next, initiating a new electrical signal in the target cell.
Major region of the diencephalon that is responsible for relaying information between the cerebrum and the hindbrain, spinal cord, and periphery.
Fleshy external structure of the ear.
Ear drum.
Structures on the lateral surface of the head, including the auricle and the ear canal back to the tympanic membrane.
Space within the temporal bone between the ear canal and bony labyrinth where the ossicles amplify sound waves from the tympanic membrane to the oval window.
Three small bones in the middle ear.
Membrane at the base of the cochlea where the stapes attaches, marking the beginning of the scala vestibuli.
Structure within the temporal bone that contains the sensory apparati of hearing and balance.
Region of the conducting zone that forms a tube of skeletal muscle lined with respiratory epithelium; located between the nasal conchae and the esophagus and trachea.
(Also pharyngotympanic tube) tube linking the pharynx and middle ear, important for equalizing air pressure in the middle ear.
An air space in the tympanic bone, posterior to (and connected with) the middle ear.
Large bony prominence on the inferior, lateral skull, just behind the earlobe.
Paired bones that form the lateral, inferior portions of the skull, with squamous, mastoid, and petrous portions.
Structure within the temporal bone that contains the sensory apparati of hearing and balance.
Auditory portion of the inner ear containing structures to transduce sound stimuli.
In the ear, the portion of the inner ear responsible for the sense of equilibrium.
Eighth cranial nerve; responsible for the sensations of hearing and balance.
Location of neuronal cell bodies that transmit auditory information along the eighth cranial nerve.
Auditory portion of the inner ear containing structures to transduce sound stimuli.
Portion of the cochlea that extends from the oval window to the apex.
Extracellular fluid of the inner ear, lying between the membranous labyrinth and the bony labyrinth.
(Also scala media) space within the auditory portion of the inner ear that contains the organ of Corti and is adjacent to the scala tympani and scala vestibuli on either side.
Portion of the cochlea that extends from the apex to the round window.
Membrane that marks the end of the scala tympani.
Three small bones in the middle ear.
Membrane separating the cochlear duct from the scala vestibuli.
In the ear, the floor of the cochlear duct on which the organ of Corti sits.
Extracellular fluid of the inner ear, lying within the membranous labyrinth.
Structure in the cochlea in which hair cells transduce movements from sound waves into electrochemical signals.
Mechanoreceptor cells found in the inner ear that transduce stimuli for the senses of hearing and balance.
Array of apical membrane extensions in a hair cell that transduce movements when they are bent.
Component of the organ of Corti that lays over the hair cells, into which the stereocilia are embedded.
Membrane-spanning protein that has an inner pore which allows the passage of one or more substances (a form of facilitated diffusion).
Tip of the cochlea where the scale vestibuli meets the scala tympani.
Middle region of the adult brain that develops from the mesencephalon.
Half of the midbrain tectum that is responsible for aligning visual, auditory, and somatosensory spatial perceptions.
Receptor cell that transduces mechanical stimuli into an electrochemical signal.
In the ear, the portion of the inner ear responsible for the sense of equilibrium.
Modality of balance relating to position of the head with respect to gravity ("which way is up") and linear acceleration.
Structure of the inner ear responsible for transducing linear acceleration in the horizontal plane.
Structure of the inner ear responsible for transducing linear acceleration in the vertical plane.
Modality of equilibrium relating to rotational movement of the head.
Part of the membranous labyrinth contained in the semicircular canals.
Location of neuronal cell bodies that transmit equilibrium information along the eighth cranial nerve.
Region of the adult brain connected primarily to the pons that developed from the metencephalon (along with the pons) and is largely responsible for comparing information from the cerebrum with sensory feedback from the periphery through the spinal cord.
Enlargement at the base of a semicircular canal at which transduction of equilibrium stimuli takes place within the ampulla.
Gelatinous substance in the utricle and saccule of the inner ear that contains calcium carbonate crystals and into which the stereocilia of hair cells are embedded.
Layer of calcium carbonate crystals located on top of the otolithic membrane.
Change in cell membrane potential below resting potential (<-70mV).
Structures within the inner ear responsible for transducing rotational movement information.
Two-dimensional, vertical plane that divides the body or organ into right and left sides.
In the ear, the structure at the base of a semicircular canal that contains the hair cells and cupula for transduction of rotational movement of the head.
Sense organ that responds to rotational movement (dynamic equilibrium), contained in each ampulla.
That part of a cell or tissue which, in general, faces an open space.
Specialized structure within the base of a semicircular canal that bends the stereocilia of hair cells when the head rotates by way of the relative movement of the enclosed fluid.
Part of the membranous labyrinth contained in the semicircular canals.
Third cranial nerve; responsible for contraction of four of the extraocular muscles, the muscle in the upper eyelid, and pupillary constriction.
Fourth cranial nerve; responsible for contraction of one of the extraocular muscles.
Sixth cranial nerve; responsible for contraction of one of the extraocular muscles.
(In anatomy), the openings for eyes in the skull.
Membrane attached to the inner surface of the eyelids that covers the anterior surface of the cornea.
Gland lateral to the orbit that produces tears to wash across the surface of the eye.
Duct in the medial corner of the orbit that drains tears into the nasal cavity.
One of six muscles originating out of the bones of the orbit and inserting into the surface of the eye which are responsible for moving the eye.
Relating to circulation of blood.
Type of tissue that serves to hold in place, connect, and integrate the body’s organs and systems.
Division of the autonomic nervous system responsible for restful and digestive functions.
Branch of the autonomic nervous system associated with emergency systems ("fight of flight").
Receptor cell specialized to respond to light stimuli.
Change in cell membrane potential below resting potential (<-70mV).
Chemical signal that is released from the synaptic end bulb of a neuron to cause a change in the target cell.
Change in voltage of a cell membrane in response to a stimulus that results in transmission of an electrical signal; unique to neurons and muscle fibres.
One of six muscles originating out of the bones of the orbit and inserting into the surface of the eye which are responsible for moving the eye.
Becoming wider, larger or more open.
Describes the middle or direction toward the middle of the body.
Describes the side or direction toward the side of the body.
The direction towards the head.
Describes a position below or lower than another part of the body proper; near or toward the tail (in humans, the coccyx, or lowest part of the spinal column); also referred to as caudal.

